8
32
P. E. Gordon, A. J. Fry / Tetrahedron Letters 42 (2001) 831–833
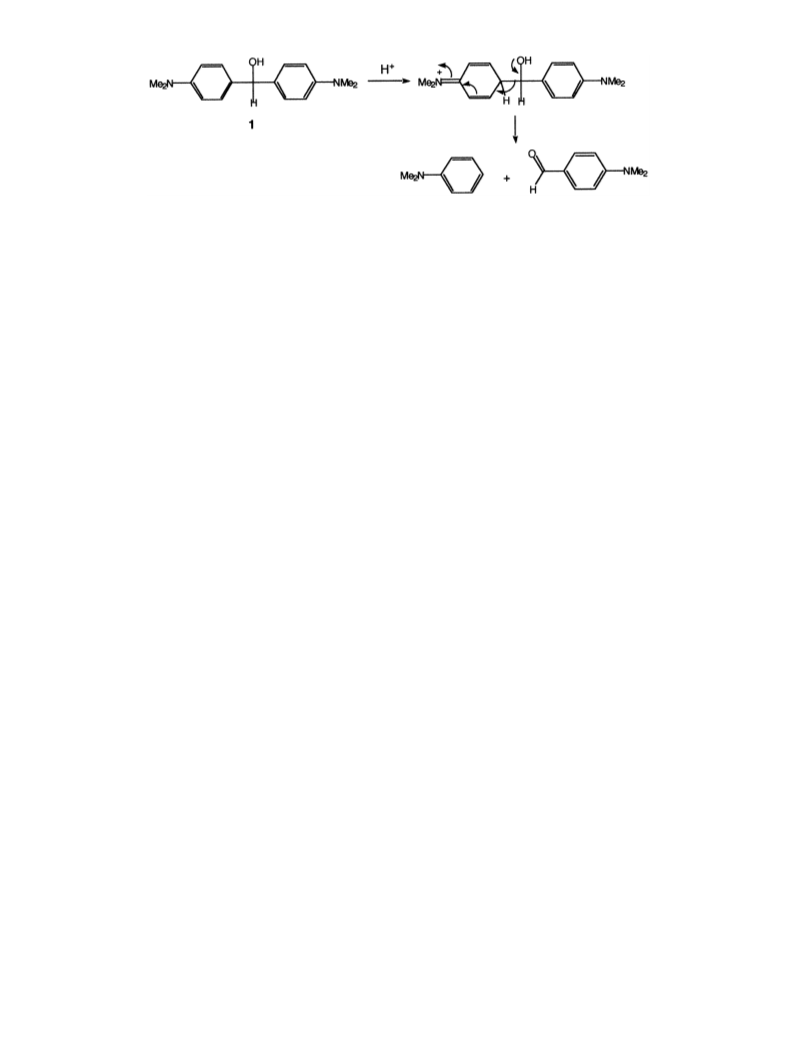
A competing reaction during the attempted reduction
of 4,4%-bis[dimethylamino]benzhydrol was cleavage to a
mixture of 4-dimethylaminobenzaldehyde and dimethy-
laniline (run 8), presumably by hydrogen iodide; an old
report showed that the same cleavage can be effected by
hydrogen bromide. The reaction is curious in that it is
most readily understood as proceeding through initial
ring protonation of 1, presumably facilitated by the
para-dimethylamino group (Scheme 1).
Reduction of 4-methylbenzhydrol (representative reac-
tion): Iodine (1.00 g; 4.0 mmol), 4-methylbenzhydrol
(0.79 g; 4.03 mmol), and acetic acid (25 mL) were
stirred under nitrogen in a flask equipped with con-
denser. Hypophosphorous acid (50% aq.; 2 mL; 19.3
mmol) was added and the mixture was heated to 60°C.
The mixture was stirred for 24 h, diluted with water,
and extracted with hexane. The hexane was dried over
1
9
MgSO and removed under reduced pressure. Analysis
4
by GC–MS showed only a single constituent, 4-
methyldiphenylmethane, produced in 100% yield. H
Selectivity: We previously reported that reduction of
substituted benzophenones to the corresponding diaryl-
methanes can be effected in high yield by this same
mixture of hypophosphorous acid and iodine in reflux-
20
1
NMR (300 MHz, CDCl : l 2.49 (s, 3H), 4.12 (s, 2H),
3
7
.15–7.54 (m, 9H).
9
ing acetic acid. We hypothesized that those reductions
All of the diphenylmethanes prepared in this study are
take place via the benzhydrol as an intermediate. How-
ever, benzhydrols were never observed when the reac-
tions were sampled throughout their progress. The
reason for this is now clear: benzhydrols are reduced
much faster than benzophenones and can in fact be
reduced at much lower temperature. When an equimo-
lar mixture of benzophenone and 3,4-dimethylbenzhy-
drol was allowed to react for as long as 72 h at 60°C,
the alcohol was completely converted to 3,4-
dimethyldiphenylmethane without any detectable
reduction of benzophenone to diphenylmethane! Simi-
lar results were obtained using mixtures of benzophe-
none with 4,4%-di-X-benzhydrol, where X is chloro,
methoxy, or dimethylamino. Reduction of substituted
benzhydrols can be carried out at temperatures as low
as 40°C, although at the expense of longer reaction
times.
known compounds except for 3,4%-bis-[trifluoromethyl]-
diphenylmethane (mp 41–42°C), which exhibited the
1
following spectral properties: H NMR (CDCl ) l 7.3–
7
3
+
.6 (m, 8H), 4.18 (s, 2H); MS (EI) m/z 304 (M ), 285
(
M−F), 264 (M−2F), 235 (M−CF , 100), 215, 165.
3
Anal. calcd for C H F: C, 59.22; H, 3.31; F, 37.47.
15 15
Found: C, 59.08; H, 3.39; F, 37.18.
Acknowledgements
Financial support by the National Science Foundation
(Grant No. CHE-97-13306 to A. J. F.) is gratefully
acknowledged. Paul Gordon received a Hughes Foun-
dation Summer Research Grant.
a
Table 1. Reduction of aryl carbinols with hypophosphorous acid–iodine in acetic acid
Run
Alcohol
Product(s), yield
1
2
3
4
5
Benzhydrol
Diphenylmethane, 100%
4-Methyldiphenylmethane, 100%
4-Hydroxydiphenylmethane, 95%; 4-acetoxydiphenylmethane, 5%
4-Bromodiphenylmethane, 100%
4-Chlorodiphenylmethane, 100%
3-Trifluoromethyldiphenylmethane, 100%
4-Trifluoromethyldiphenylmethane, 100%
4,4%-Bis[dimethylamino]diphenylmethane, 50%; 4-dimethylaminobenzaldehyde, 50%
4-Methylbenzhydrol
4-Methoxybenzhydrol
4-Bromobenzhydrol
4-Chlorobenzhydrol
3-Trifluoromethylbenzhydrol
4-Trifluoromethylbenzhydrol
4,4%-Bis[dimethylamino]-
benzhydrol
b
6
7
b
8
9
3,4-Dimethylbenzhydrol
4,4%-Dimethoxybenzhydrol
4,4%-Dichlorobenzhydrol
4,4%-Diiodobenzhydrol
3,4-Dimethyldiphenylmethane, 100%
4,4%-Dimethoxydiphenylmethane, 100%
4,4%-Dichlorodiphenylmethane, 100%
Diphenylmethane, 65%; 4-iododiphenylmethane, 19%; 4,4%-diiododiphenylmethane, 8%;
benzhydrol, 3.5%; 4-iodobenzhydrol, 3.5%; 4,4%-diiodobenzhydrol, 1%
3,3%-Bis[trifluoromethyl]diphenylmethane, 100%
1
1
1
0
1
2
1
1
3b
3,3%-Bis[trifluoromethyl]-
benzhydrol
3,4%-Bis[trifluoromethyl]-
benzhydrol
4b
3,4%-Bis[trifluoromethyl]diphenylmethane, 100%
1
1
1
5
6
7
4,4%-Dimethylbenzhydrol
Triphenylmethanol
1,1-Diphenylethanol
4,4%-Dimethyldiphenylmethane, 100%
Triphenylmethane, 100%
1,1-Diphenylethane, 100%
a
Alcohol (4.0 mmol), iodine (0.4 mmol), 50% aq. hypophosphorous acid (19.3 mmol), acetic acid (25 mL), 24 h under nitrogen at 60°C.
Complete reaction required 48–72 h.
b

 Gordon, Paul E.
Gordon, Paul E.
 Fry, Albert J.
Fry, Albert J.