CHEMPLUSCHEM
FULL PAPERS
DOI: 10.1002/cplu.201300114
Oxidation of Toluene and Other Examples of CÀH Bond
Activation by CdO2 and ZnO2 Nanoparticles
S. R. Lingampalli, Uttam Gupta, Ujjal K. Gautam,* and C. N. R. Rao*[a]
Nanoparticles of CdO2 and ZnO2 are shown to oxidize toluene
primarily to benzaldehyde in the 160–1808C range, around
which temperature the nanoparticles decompose to give the
oxides. The product selectivity and other features of the reac-
tion are explained taking into account the various byproducts
formed in the reaction. These metal peroxides also activate the
CÀH bonds of cyclohexane. It is possible to bring down the re-
action temperature by partially substituting the Zn in ZnO2
with Ni.
Introduction
Activation of the CÀH bond has been getting increasing atten-
tion in the last few years.[1–4] Alkyl aromatic compounds, such
as toluene, constitute an important family of compounds and
a reaction of importance in this context is the oxidation of tol-
uene to benzaldehyde and benzyl alcohol.[5–12] Commercially,
benzaldehyde is produced either as a byproduct during the
conversion of toluene to benzoic acid by aerial oxidation or at
times, by the halogenation of toluene followed by saponifica-
tion.[13] The main concern in these reactions is the control of
the product distribution, which is nontrivial. Recently,
a number of strategies have been explored that employ mo-
lecular oxygen or other oxidizing agents.[14–17] Heterogeneous
catalysis, which is advantageous owing to the ease of separa-
tion of the catalysts, has also been employed for this purpose.
Thus, Raja et al.[18] designed single-site heterogeneous catalysts
for the solvent-free oxidation of toluene using aluminophos-
phates. Hutchings and co-workers[5] have recently reported an
excellent method for the oxidation of toluene by molecular
oxygen under high pressure and at approximately 1658C, in
the presence of catalytic Au–Pd alloy nanoparticles. This
method is solvent-free, does not require post-reaction solvent
separation, and yields benzyl benzoate in excess of 85%. The
use of H2O2 and organic peroxides as sources of oxygen for
the oxidation of toluene has also been reported.[16,19] Another
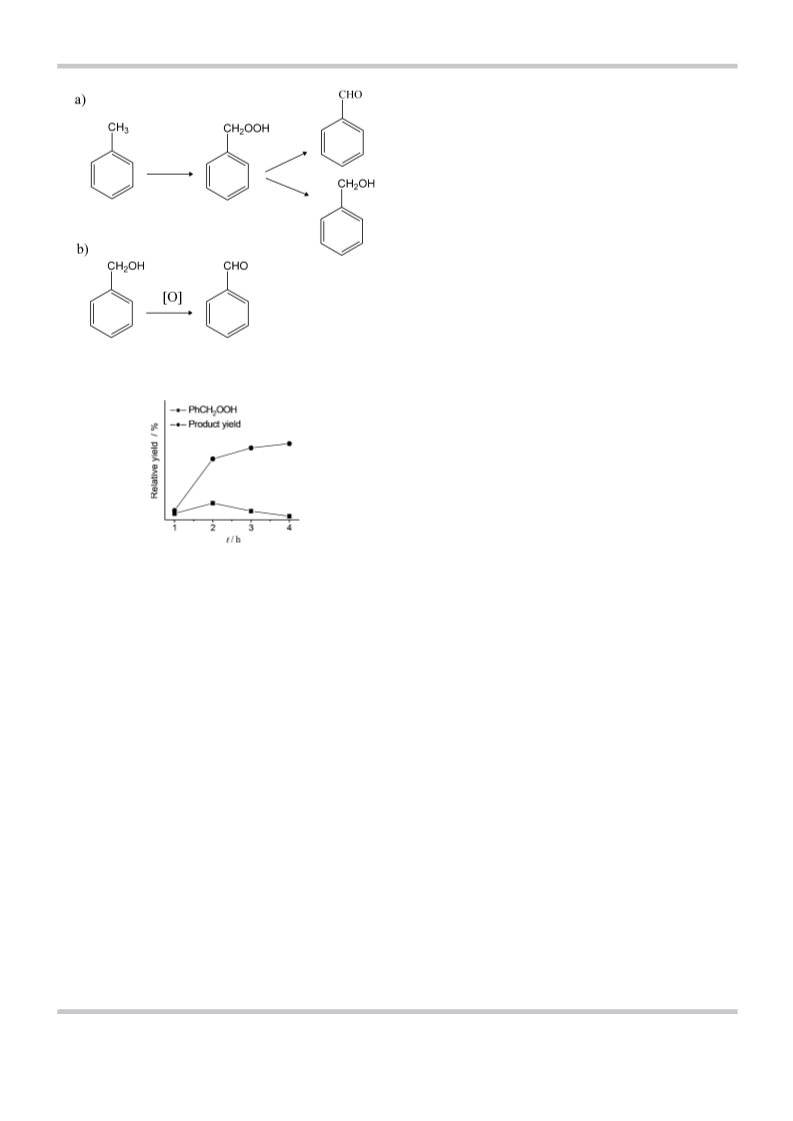
important reaction involving CÀH activation is the oxidation of
cyclohexane to cyclohexanol and cyclohexanone.[20,21]
cohol. We made use of nanoparticles of CdO2 and ZnO2 for the
purpose because they decompose at relatively lower tempera-
tures, well below the decomposition temperatures of stable
metal peroxides, such as MgO2 and BaO2,[22–25] to give oxygen
and the metal oxide. Furthermore, surfaces of metal peroxides
are known to contain reactive species, such as peroxy or hy-
droperoxy radicals, with the potential to initiate radical chain
reactions and to activate CÀH bonds.[26,27] Cleavage of the
peroxy bonds even at low temperatures, as reported in the
case of hydrogen peroxide, leads to reactive singlet oxygen
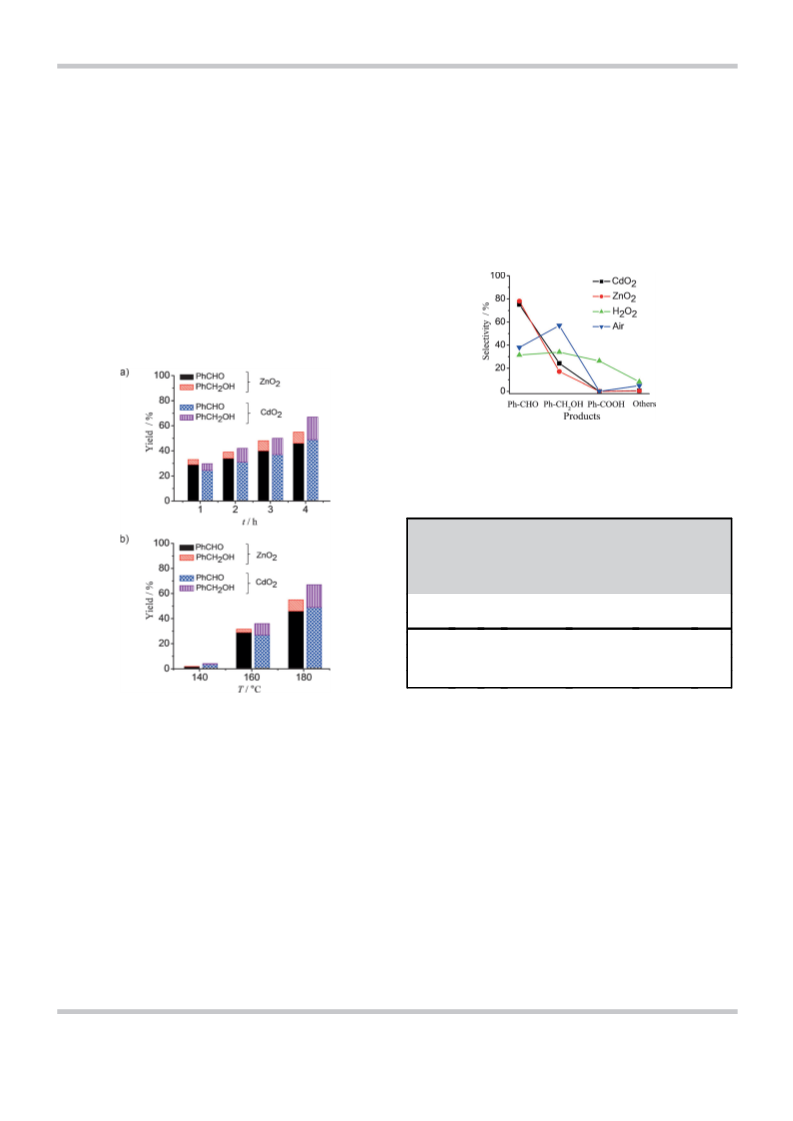
species.[28] Besides demonstrating the facile oxidation of tolu-
ene by solid CdO2 and ZnO2 nanoparticles in the 160–1808C
range, which primarily yields benzaldehyde, we show that the
ZnÀO surface influences product selectivity. We performed pre-
liminary studies on the oxidation of cyclohexane by the perox-
ide nanoparticles to examine their general use in CÀH bond
activation. In addition to being solvent-free, the metal peroxide
route seems to have certain advantages.
Nanoparticles of ZnO2 and CdO2 were prepared by the reac-
tion of ZnO and CdO with excess H2O2 at 808C (1 h) and
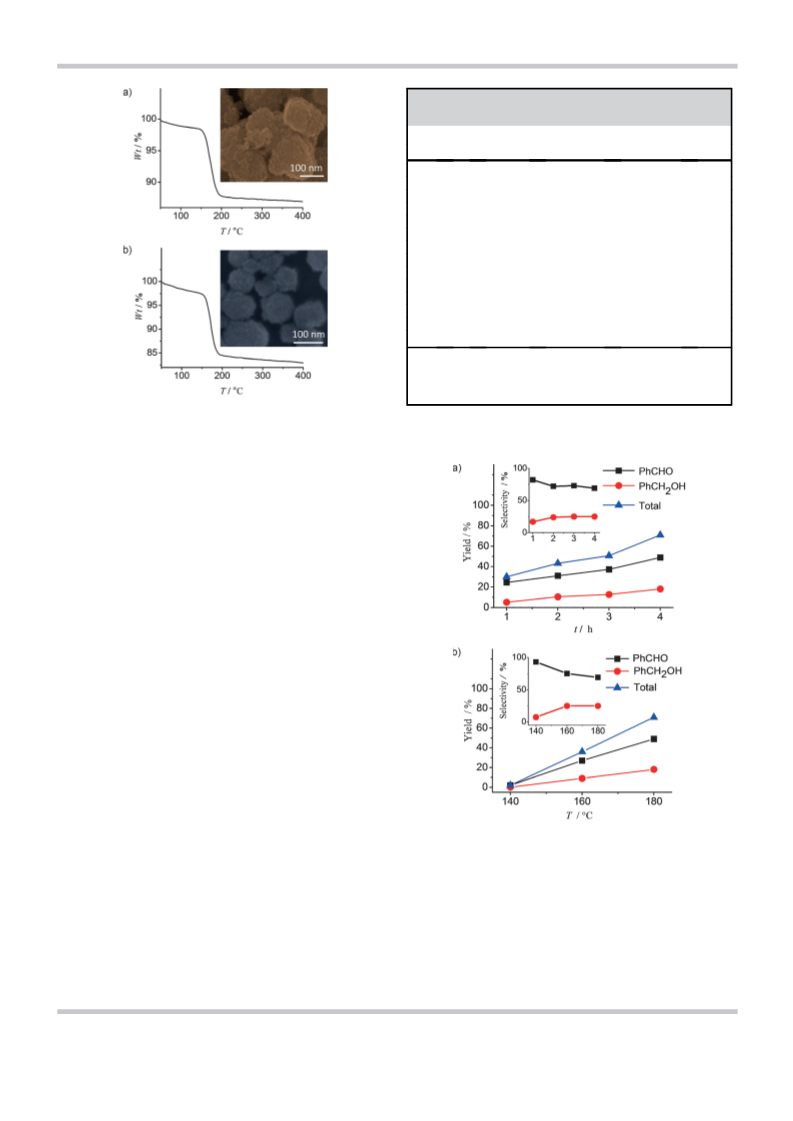
1008C (12 h), respectively. The peroxide particles so obtained
are crystalline with diameters in the 5–10 nm range (see Sup-
porting Information, Figure S1). Importantly, these reaction
conditions are mild in comparison with many other known
synthetic procedures.[29,30] The BET surface areas of the ZnO2
and CdO2 particles were 54 and 46 m2 gÀ1, respectively. The de-
composition temperatures of the peroxides were in the range
160–1808C as revealed by thermogravimetric analysis
(Figure 1). In a typical experiment to oxidize toluene, a prede-
termined amount of toluene and metal peroxide were sealed
in a Teflon-lined autoclave and heated at the desired tempera-
ture for a fixed period of time in a preheated air oven. The
product obtained was characterized by GC–MS and NMR spec-
troscopy.
Herein, we report the oxidation of toluene by metal perox-
ides mainly to benzaldehyde, with a small fraction of benzyl al-
[a] S. R. Lingampalli, U. Gupta, Dr. U. K. Gautam, Prof. C. N. R. Rao
New Chemistry Unit
CSIR Centre of Excellence in Chemistry and Sheikh Saqr Laboratory
Jawaharlal Nehru Centre for Advanced Scientific Research
Jakkur P.O., Bangalore 560064 (India)
Fax: (+91)80-2208-2760
Supporting information for this article is available on the WWW under
ꢀ 2013 Wiley-VCH Verlag GmbH & Co. KGaA, Weinheim
ChemPlusChem 0000, 00, 1 – 7
&
2
&
ÞÞ
These are not the final page numbers!

 Lingampalli
Lingampalli
 Gupta, Uttam
Gupta, Uttam
 Gautam, Ujjal K.
Gautam, Ujjal K.
 Rao
Rao