G. Sartori et al. / Journal of Catalysis 222 (2004) 410–418
417
the rate-determining step could be the β-elimination step,
which regenerates the amino function on the catalyst.
finement of the amine groups in unaccessible environments.
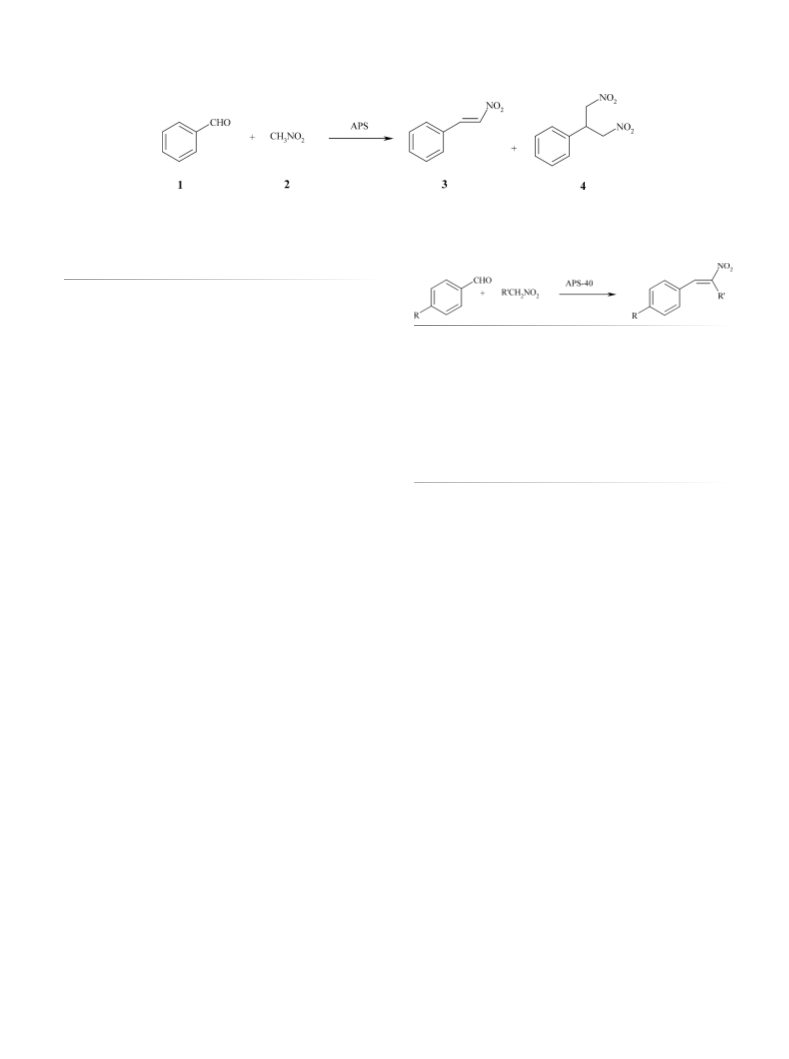
The reactivity results exhibit a similar trend.
With the best catalyst, APS-40, (E)-nitrostyrene was thus
obtained in high yield (95%) and selectivity (97%) under
mild heterogeneous conditions. The reaction was extended
to nitroethane and to different aldehydes, suggesting a pecu-
liar reaction mechanism.
3
.3. Leaching test and recycling
To exclude the possible leaching of any active catalytic
species into solution, the model reaction between benzalde-
hyde and nitromethane was examined by following the stan-
dard procedure suggested by Lempers and Sheldon [29].
Acknowledgments
◦
Thus, the reaction mixture was filtered at 90 C after 30 min
(
when product 3a was produced in 60% yield) and the fil-
The authors acknowledge the support of the Ministero
dell’Università e della Ricerca Scientifica e Tecnologica
◦
trate was further heated at 90 C for 2 h. Product 3a was
detected in 65% total yield (60%+5%). In contrast, addition
(
(
MURST), Italy, the Consiglio Nazionale delle Ricerche
CNR), Italy, and the University of Parma (National Project
of both benzaldehyde (5 mmol, 0.53 g) and nitromethane
◦
(
6 mL) to the recovered solid catalyst and heating at 90 C
“Processi Puliti per la Chimica Fine”). D.J.M. thanks the
for 2 h afforded nitrostyrene 3a in 90% yield. These results
confirm that the reaction actually occurs on the supported
propylamine.
Royal Society for a University Research Fellowship.
The authors are grateful to the Centro Interdipartimentale
Misure (CIM) for the use of NMR and mass instruments and
to Mr. Pier Antonio Bonaldi (Lillo) for technical collabora-
tion.
The recycling of the APS-40 catalyst was conducted by
◦
filtering, washing with methylene chloride, drying at 90 C
for 2 h (under vacuum), and immediately reusing without
any activation. On reuse, the catalyst suffered some deacti-
vation giving compound 3ax in 95, 90, 60, and 25% yields
in four cycles.
Finally, authors at the Dipartimento di Chimica Or-
ganica e Industriale of Parma and at the Dipartimento di
Chimica IFM of Torino participated in this work within the
framework of the activity of the Interuniversity Consortium
To understand the catalyst deactivation we reacted ben-
zaldehyde with nitromethane over APS-40 in the presence
of an equimolecular amount of nitrostyrene. Product 3ax
was obtained in 80% yield after 5 h under these conditions,
suggesting that the catalyst is still active, but at a lower
reaction rate. Moreover, IR investigation of the reused cat-
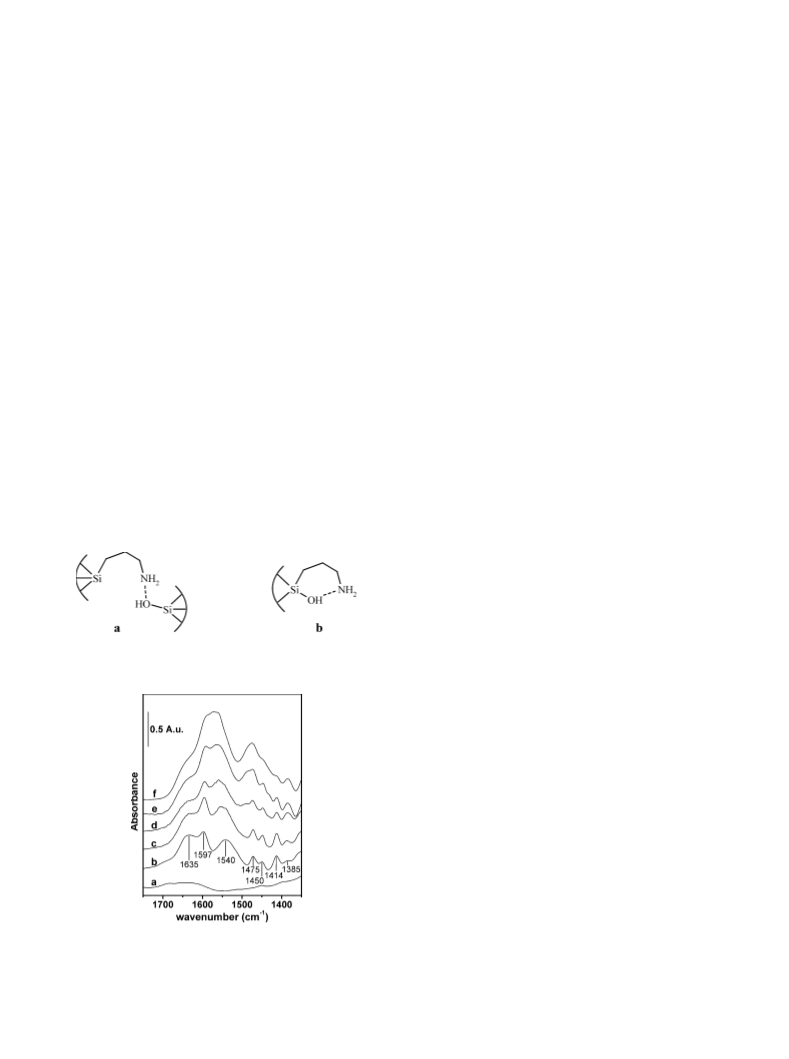
alyst indicated the presence of strong bands at 1635, 1540,
“
Chemistry for the Environment”—INCA.
References
[
[
[
1] A.R. Sheldon, Chem. Ind. (London) (1997) 12.
2] C. Adams, Chem. Ind. (London) (1999) 740.
3] J.H. Clark, D.J. Macquarrie, Chem. Soc. Rev. (1996) 303.
−1
ꢀ
+
and 1414 cm corresponding to the pattern of R – NH3 –
[4] G. Rosini, in: B.M. Trost, I. Fleming, C.H. Heathcock (Eds.), Com-
prehensive Organic Synthesis, Pergamon, Oxford, 1991, p. 321.
[
[
[
−
ꢀꢀ
OOCR salt, probably due to reaction of the supported
5] R. Ballini, G. Bosica, J. Org. Chem. 62 (1997) 425.
6] R. Ballini, G. Bosica, P. Forconi, Tetrahedron 52 (1996) 1677.
7] R. Ballini, F. Bigi, E. Gogni, R. Maggi, G. Sartori, J. Catal. 191 (2000)
348.
8] G. Sartori, F. Bigi, R. Maggi, A. Mazzacani, G. Oppici, Eur. J. Org.
Chem. (2001) 2513.
propylamine with CO2 present in the air, or with some ben-
zoic acid present in the benzaldehyde or produced by in situ
oxidation, the reaction not being performed in a controlled
atmosphere.
[
To regenerate the propylamine active sites, the reused
catalyst was treated with a 10% ethanolic solution of propyl-
amine and then washed with ethanol in a Soxhlet apparatus
[
9] S. Carloni, D.E. De Vos, P.A. Jacobs, R. Maggi, G. Sartori, R. Sartorio,
J. Catal. 205 (2002) 199.
[
[
[
[
10] G. Sartori, A. Armstrong, R. Maggi, A. Mazzacani, R. Sartorio, F. Bi-
gi, B. Dominguez-Fernandez, J. Org. Chem. 68 (2003) 3232.
11] D.J. Macquarrie, R. Maggi, A. Mazzacani, G. Sartori, R. Sartorio,
Appl. Catal. A 246 (2003) 183.
12] V.J. Bulbule, V.H. Deshpande, S. Velu, A. Sudalai, S. Sivasankar, V.T.
Sathe, Tetrahedron 55 (1999) 9325.
◦
for 4 h. After being dried at 110 C for 2 h the catalyst was
tested in the model reaction affording product 3ax in 78%
yield and finally showing that most of the active sites could
be regenerated by convenient basic treatment.
13] G. Demicheli, R. Maggi, A. Mazzacani, P. Righi, G. Sartori, F. Bigi,
Tetrahedron Lett. 42 (2001) 2401.
4
. Conclusions
[14] M. Lakshmi Kantam, P. Streekanth, Catal. Lett. 57 (1999) 227.
[15] L.L. Hench, J.K. West, Chem. Rev. 90 (1990) 33.
[
16] E.I. Ko, in: G. Erdtl, H. Knozinger, J. Weitkamp (Eds.), Handbook of
Heterogeneous Catalysis, Pergamon, Oxford, 1997, p. 321.
17] P.A. Webb, C. Orr, Analytical Methods in Fine Particle Technology,
Micromeritics Instruments Corporation, Norcross, USA, 1997.
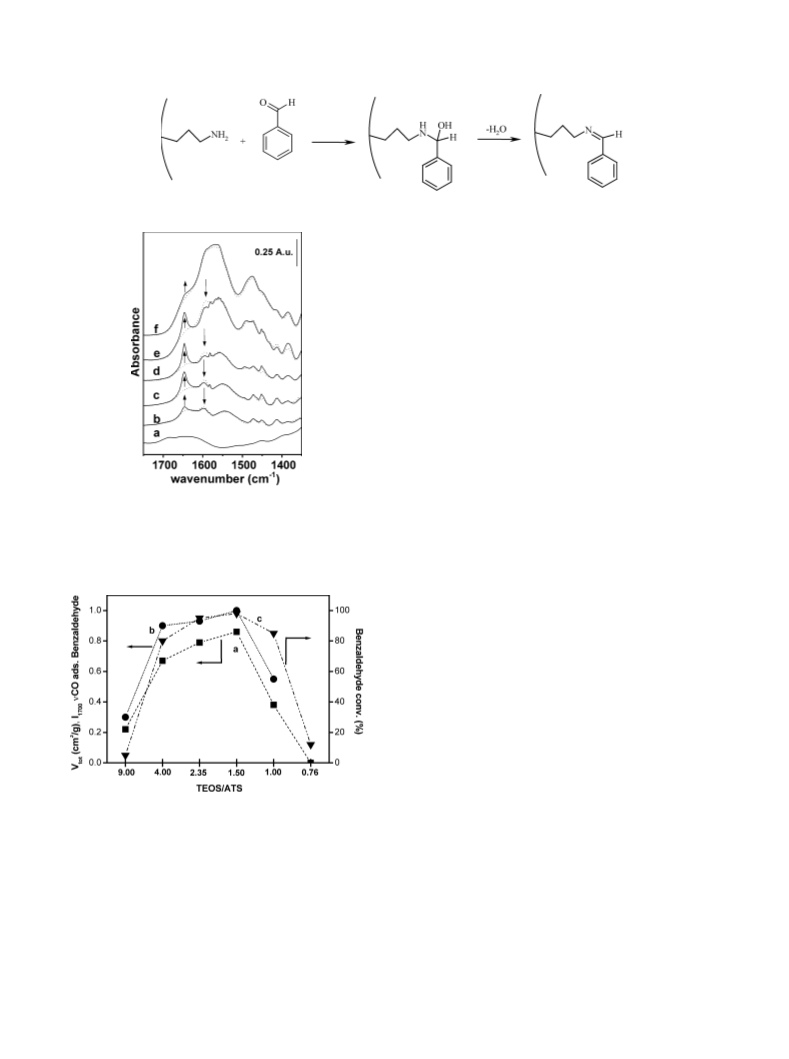
The catalytic activity of the different APS materials pre-
pared by sol–gel technique depends on the physicochemical
properties of the solids which are conveniently tuned by
changing the relative ratio of the starting sol components
TEOS/ATS. Studies of the textural and adsorption proper-
ties of the APS materials indicated that increasing the amine
percentage over 40% in the gel synthesis causes a collapse
of the surface area and pore volume of the solids, with con-
[
[18] F. Roquerol, J. Roquerol, K. Sing, Adsorption by Powders and Porous
Solids, Academic Press, UK, 1999.
[19] A. Burneau, J.P. Gallas, in: A.P. Legrand (Ed.), The Surface Properties
of Silicas, Wiley, Chichester, 1998, p. 147.
[20] N.B. Colthup, L.H. Daly, S.E. Wiberley, Introduction to Infrared and
Raman Spectroscopy, 2nd ed., Academic Press, New York, 1975.

 Sartori
Sartori
 Bigi
Bigi
 Maggi
Maggi
 Sartorio
Sartorio
 Macquarrie
Macquarrie
 Lenarda
Lenarda
 Storaro
Storaro
 Coluccia
Coluccia
 Martra
Martra