Journal of the American Chemical Society
Page 10 of 12
solvent serve as an important co-catalyst which is responsible
for the activation of the substrate. Control experiments clearly
showed that the synergistic interplay between the proton and
the supramolecular capsule is essential for the successful tail-
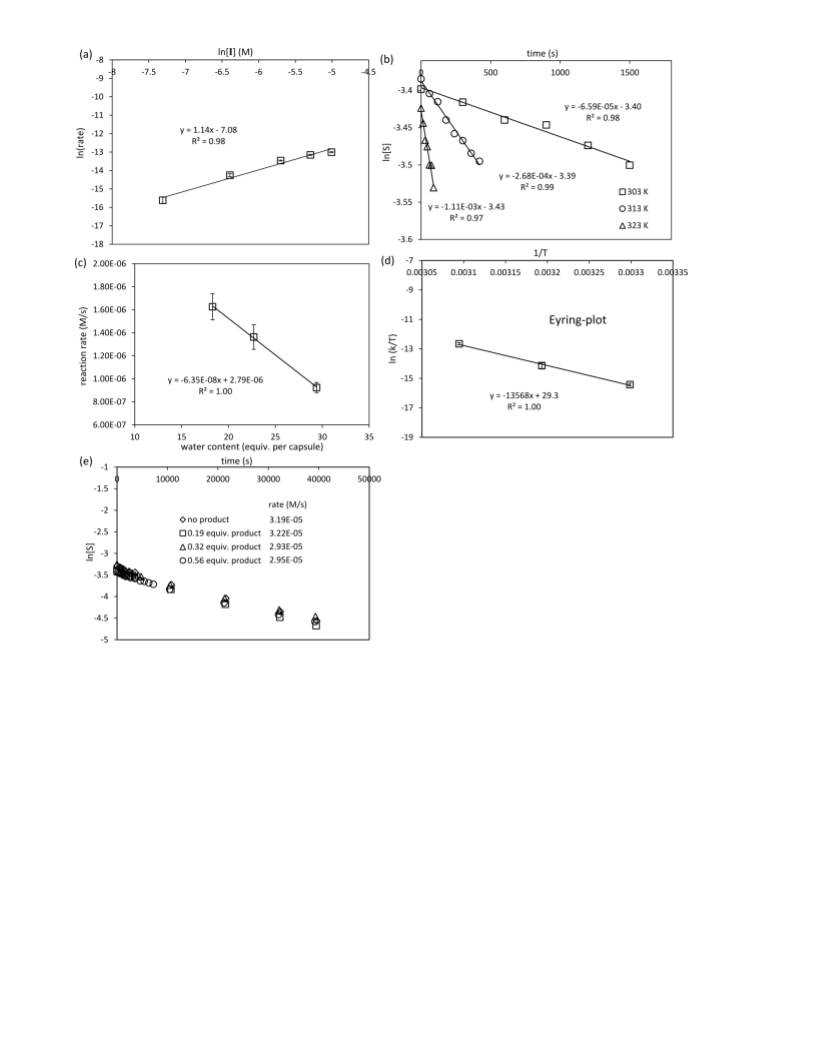
to-head terpene cyclization. Kinetic analysis suggests that,
under the standard reaction conditions, the cleavage of the
leaving group is the rate-determining step. In water-saturated
chloroform, however, protonation of the bound substrate is the
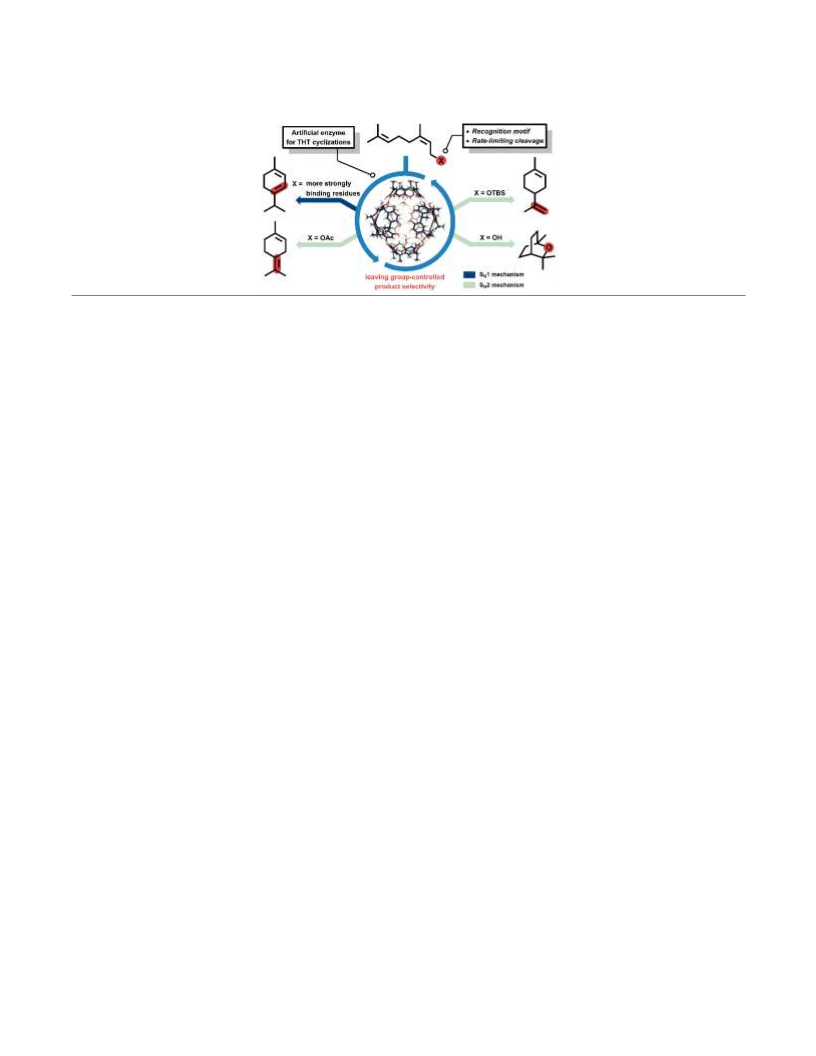
slowest step. These studies demonstrate that a relatively sim-
ple aromatic cavity is capable of catalyzing the challenging
tail-to-head terpene cyclization reaction in a selective fashion.
Synthesis of more complex terpene natural products utilizing
this strategy is currently ongoing in our laboratories.
Leenders, S. H. A. M.; Gramage-Doria, R.; de Bruin, B.; Reek, J. N.
H., Chem. Soc. Rev. 2015,44, 433-448.
. (a) Rebek, J., Acc. Chem. Res. 2009,42, 1660-1668; (b) Ajami, D.;
Rebek, J., Acc. Chem. Res. 2012,46, 990-999; (c) Ajami, D.; Liu, L.;
Rebek Jr, J., Chem. Soc. Rev. 2015,44, 490-499.
1
2
3
4
5
6
7
8
9
1
1
1
1
1
1
1
1
1
1
2
2
2
2
2
2
2
2
2
2
3
3
3
3
3
3
3
3
3
3
4
4
4
4
4
4
4
4
4
4
5
5
5
5
5
5
5
5
5
5
6
9
1
0.Jordan, J. H.; Gibb, B. C., Chem. Soc. Rev. 2014,44, 547-585.
11.(a) Yoshizawa, M.; Klosterman, J. K.; Fujita, M., Angew. Chem. Int.
Ed. 2009,48, 3418-3438; (b) Meeuwissen, J.; Reek, J. N. H., Nat
Chem 2010,2, 615-621; (c) Marchetti, L.; Levine, M., ACS Catal.
2011,1, 1090-1118; (d) Wiester, M. J.; Ulmann, P. A.; Mirkin, C. A.,
Angew. Chem. Int. Ed. 2011,50, 114-137; (e) Raynal, M.; Ballester, P.;
Vidal-Ferran, A.; van Leeuwen, P. W. N. M., Chem. Soc. Rev. 2014,43,
0
1
2
3
4
5
6
7
8
9
0
1
2
3
4
5
6
7
8
9
0
1
2
3
4
5
6
7
8
9
0
1
2
3
4
5
6
7
8
9
0
1
2
3
4
5
6
7
8
9
0
1
734-1787; (f) Brown, C. J.; Toste, F. D.; Bergman, R. G.; Raymond,
K. N., Chem. Rev. 2015,115, 3012-3035; (g) Zarra, S.; Wood, D. M.;
Roberts, D. A.; Nitschke, J. R., Chem. Soc. Rev. 2015,44, 419-432; (h)
Catti, L.; Zhang, Q.; Tiefenbacher, K., Synthesis 2016,48, 313-328; (i)
Catti, L.; Zhang, Q.; Tiefenbacher, K., Chem. Eur. J. 2016,22, 9060-
9066.For recent examples see:(j) Cullen, W.; Misuraca, M. C.; Hunter,
C. A.; Williams, N. H.; Ward, M. D., Nat. Chem.2016,8, 231-236; (k)
Levin, M. D.; Kaphan, D. M.; Hong, C. M.; Bergman, R. G.;
Raymond, K. N.; Toste, F. D., J. Am. Chem. Soc. 2016,138, 9682-
ASSOCIATED CONTENT
Supporting Information. This material is available free of
charge via the Internet at http://pubs.acs.org.
AUTHOR INFORMATION
Corresponding Author
9
693; (l) Jans, A. C. H.; Gómez-Suárez, A.; Nolan, S. P.; Reek, J. N.
H., Chem. Eur. J. 2016,22, 14836-14839; (m) Kuijpers, P. F.; Otte, M.;
Dürr, M.; Ivanović-Burmazović, I.; Reek, J. N. H.; de Bruin, B., ACS
Catal. 2016,6, 3106-3112; (n) Wang, Q.-Q.; Gonell, S.; Leenders, S.
H. A. M.; Dürr, M.; Ivanović-Burmazović, I.; Reek, J. N. H., Nat.
Chem.2016,8, 225-230; (o) Ueda, Y.; Ito, H.; Fujita, D.; Fujita, M., J.
Am. Chem. Soc. 2017, asap.
konrad.tiefenbacher@unibas.ch / tkonrad@ethz.ch
Notes
The authors declare no competing financial interest.
1
1
2
2.(a) Zhang, Q.; Tiefenbacher, K., J. Am. Chem. Soc. 2013,135,
6213-16219; (b) Catti, L.; Tiefenbacher, K., Chem. Commun.
015,51, 892-894; (c) Zhang, Q.; Tiefenbacher, K., Nat. Chem.
ACKNOWLEDGMENT
This work was supported by funding from the European Research
Council Horizon 2020 Programme [ERC Starting grant 714620-
TERPENECAT], the Swiss National Science Foundation as part
of the NCCR Molecular Systems Engineering and the Bayerische
Akademie der Wissenschaften (Junges Kolleg). We thank Prof.
Dr. Wolfgang Eisenreich (Technische Universität München) for
2015,7, 197-202; (d) Bräuer, T. M.; Zhang, Q.; Tiefenbacher, K.,
Angew. Chem. Int. Ed. 2016,55, 7698-7701; (e) Catti, L.; Pöthig, A.;
Tiefenbacher, K., Adv. Synth. Catal. 2017,359, 1331-1338.
1
3.(a) Bianchini, G.; La Sorella, G.; Canever, N.; Scarso, A.; Strukul,
G., Chem. Commun. 2013,49, 5322-5324; (b) Giust, S.; La Sorella, G.;
Sperni, L.; Fabris, F.; Strukul, G.; Scarso, A., Asian J. Org. Chem.
2015,4, 217-220; (c) La Sorella, G.; Sperni, L.; Strukul, G.; Scarso, A.,
ChemCatChem 2015,7, 291-296; (d) Caneva, T.; Sperni, L.; Strukul,
G.; Scarso, A., RSC Adv. 2016,6, 83505-83509; (e) Caneva, T.; Sperni,
L.; Strukul, G.; Scarso, A., RSC Adv. 2016,6 (87), 83505-83509; (f)
La Sorella, G.; Sperni, L.; Ballester, P.; Strukul, G.; Scarso, A., Catal.
Sci. Technol. 2016,6, 6031-6036; (g) La Sorella, G.; Sperni, L.;
Strukul, G.; Scarso, A., Adv. Synth. Catal. 2016,358, 3443 –3449.
14.(a) MacGillivray, L. R.; Atwood, J. L., Nature 1997,389, 469-472;
(b) Avram, L.; Cohen, Y., J. Am. Chem. Soc. 2002,124, 15148-15149;
(c) Avram, L.; Cohen, Y.; Rebek, J., Chem. Commun. 2011,47, 5368-
5375.
15.Yamanaka, M.; Shivanyuk, A.; Rebek, J., J. Am. Chem. Soc.
2004,126, 2939-2943.
16.Shivanyuk, A.; Rebek, J., Proc. Natl. Acad. Sci. U.S.A. 2001,98,
7662-7665.
17.(a) Leão Lana, E. J.; da Silva Rocha, K. A.; Kozhevnikov, I. V.;
Gusevskaya, E. V., J. Mol. Catal. A: Chem. 2006,259, 99-102; (b)
Kelly, B. D.; Allen, J. M.; Tundel, R. E.; Lambert, T. H., Org. Lett.
2009,11, 1381-1383.
18.Bugarčić, Z. M.; Dunkić, J. D.; Mojsilović, B. M., Heteroat. Chem.
2004,15, 468-470.
2
the help with H NMR measurement. Dr. Ingo Schnapperelle is
acknowledged for help with GC-MS (SIM) measurements.
REFERENCES
1
.(a) Croteau, R., Chem. Rev. 1987,87, 929-954; (b) Cane, D. E.,
Chem. Rev. 1990,90, 1089-1103; (c) Christianson, D. W., Chem. Rev.
2006,106, 3412-3442; (d) Degenhardt, J.; Köllner, T. G.; Gershenzon,
J., Phytochemistry 2009,70, 1621-1637; (e) Dickschat, J. S., Nat. Prod.
Rep. 2011,28, 1917-1936; (f) Miller, D. J.; Allemann, R. K., Nat. Prod.
Rep. 2012,29, 60-71; (g) Dickschat, J. S., Nat. Prod. Rep. 2016,33,
8
2
7-110.
.Paddon, C. J.; Westfall, P. J.; Pitera, D. J.; Benjamin, K.; Fisher, K.;
McPhee, D.; Leavell, M. D.; Tai, A.; Main, A.; Eng, D.; Polichuk, D.
R.; Teoh, K. H.; Reed, D. W.; Treynor, T.; Lenihan, J.; Jiang, H.;
Fleck, M.; Bajad, S.; Dang, G.; Dengrove, D.; Diola, D.; Dorin, G.;
Ellens, K. W.; Fickes, S.; Galazzo, J.; Gaucher, S. P.; Geistlinger, T.;
Henry, R.; Hepp, M.; Horning, T.; Iqbal, T.; Kizer, L.; Lieu, B.; Melis,
D.; Moss, N.; Regentin, R.; Secrest, S.; Tsuruta, H.; Vazquez, R.;
Westblade, L. F.; Xu, L.; Yu, M.; Zhang, Y.; Zhao, L.; Lievense, J.;
Covello, P. S.; Keasling, J. D.; Reiling, K. K.; Renninger, N. S.;
Newman, J. D., Nature 2013,496, 528-532.
19.Wise, M. L.; Urbansky, M.; Helms, G. L.; Coates, R. M.; Croteau,
R., J. Am. Chem. Soc. 2002,124, 8546-8547.
3
4
.Kingston, D. G. I., J. Nat. Prod. 2000,63, 726-734.
.Malik, S.; Cusidó, R. M.; Mirjalili, M. H.; Moyano, E.; Palazón, J.;
20.Croteau, R. B.; Shaskus, J. J.; Renstrom, B.; Felton, N. M.; Cane,
D. E.; Saito, A.; Chang, C., Biochemistry 1985,24, 7077-7085.
21.Croteau, R., Arch. Biochem. Biophys. 1986,251, 777-782.
22.Avram, L.; Cohen, Y., Org. Lett. 2003,5, 3329-3332.
23.Evan-Salem, T.; Baruch, I.; Avram, L.; Cohen, Y.; Palmer, L. C.;
Rebek, J., Proc. Natl. Acad. Sci. U.S.A. 2006,103, 12296-12300.
24.The protons greatly outnumber the added deuteron. For a
resorcinarene capsule sample with 10 equiv. water, the H:D-ratio is
99:1, As a result, the observed reaction was dominantly triggered by
protonation and therefore displayed very similar reaction rate as the
reaction where protons function as the sole activation source.
Bonfill, M., Process Biochem. 2011,46, 23-34.
5.Wendt, K. U.; Schulz, G. E., Structure 1998,6, 127-133.
6.Pronin, S. V.; Shenvi, R. A., Nat. Chem. 2012,4, 915-920.
7
8
2
.Yoder, R. A.; Johnston, J. N., Chem. Rev. 2005,105, 4730-4756.
.(a) Pluth, M. D.; Bergman, R. G.; Raymond, K. N., Acc. Chem. Res.
009,42, 1650-1659; (b) Yoshizawa, M.; Fujita, M., Bull. Chem. Soc.
Jpn. 2010,83, 609-618; (c) Ronson, T. K.; Zarra, S.; Black, S. P.;
Nitschke, J. R., Chem. Commun. 2013,49, 2476-2490; (d) Han, M.;
Engelhard, D. M.; Clever, G. H., Chem. Soc. Rev. 2014,43, 1848-1860;
(
e) Zhang, G.; Mastalerz, M., Chem. Soc. Rev. 2014,43, 1934-1947; (f)
ACS Paragon Plus Environment

 Zhang, Qi
Zhang, Qi
 Catti, Lorenzo
Catti, Lorenzo
 Pleiss, Jürgen
Pleiss, Jürgen
 Tiefenbacher, Konrad
Tiefenbacher, Konrad