Page 7 of 9
Organic & Biomolecular Chemistry
Please do not adjust margins
Journal Name
ARTICLE
mM Tris-HCl buffer, 50 mM NaCl, 1 mM DTT, 5% glycerol, pH 7.4) were identified by MS, 1H NMR and 13C NMR spectroscopic data
DOI: 10.1039/C9OB01352K
using an Amicon Ultra-30K centrifugal concentrator (Millipore, analysis (Figures S7‒S29).
USA). The protein purity was confirmed by by sodium dodecyl
sulfate-polyacrylamide gel electrophoresis (SDS-PAGE), and the
protein concentration for all of the studies was determined
using the Protein Quantitative Kit (TransGen Biotech, China).
The Spectroscopic Data of Prepared Glucuronides
Icariin (1a, isolated yield 32.8%). 1H NMR (600 MHz, DMSO-d6):
δH 12.56 (1H, s, 5-OH), 7.89 (2H, d, J=8.1 Hz, H-2′/6′), 7.13 (2H,
The final protein was flash-frozen in liquid nitrogen and stored
d, J=8.2 Hz, H-3′/5′), 6.63 (1H, s, H-6), 5.28 (1H, d, J=1.9 Hz, H-
at −80 °C. For example, the approximate protein yield for EpGT8
1′′′), 5.17 (1H, t, J=9.0 Hz, H-2′′), 5.00 (1H, d, J=9.0 Hz, H-1′′′′),
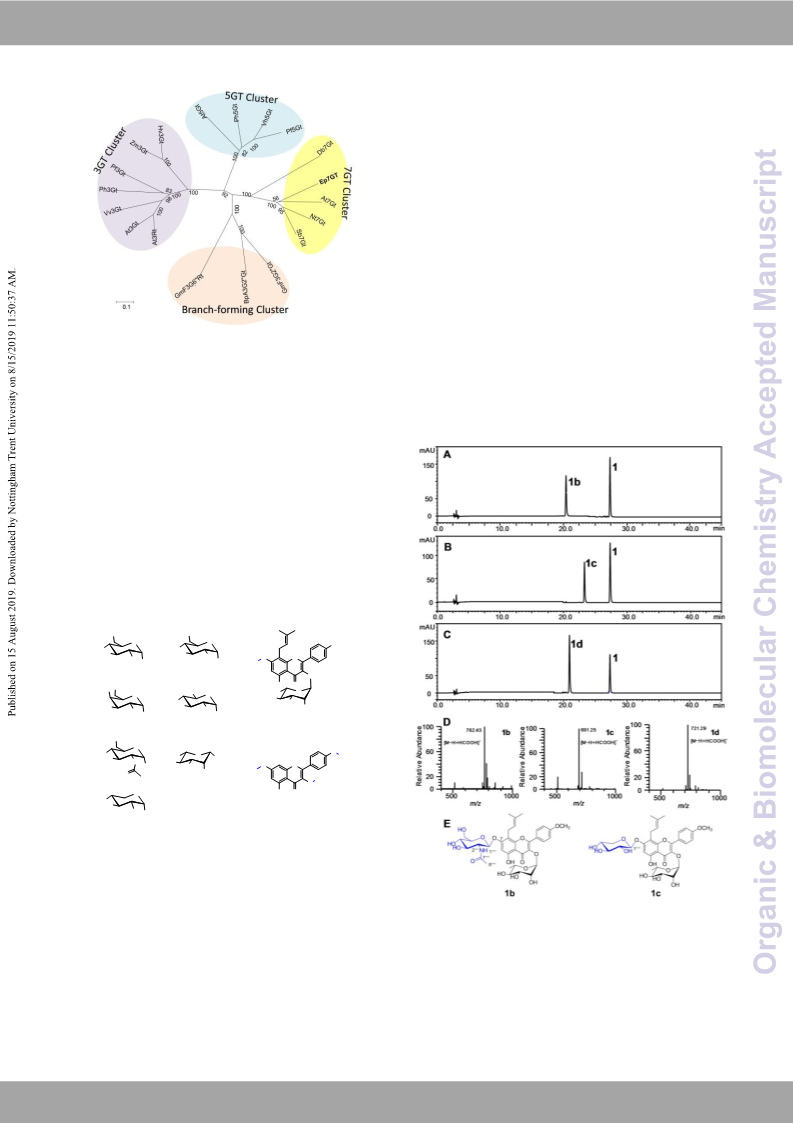
(Ep7GT) (54.1 kDa, Figure S1) was 5.4 mg/L.
3.86 (1H, s, 3H, O-CH3), 3.34 (2H, H-1′′), 3.07–4.00 (protons in
rhamnose and glucose), 1.69 (3H, s, H-4′′), 1.60 (3H, s, H-5′′),
0.79 (3H, d, J=7.2 Hz, H-6′′′); 13C NMR (125 MHz, DMSO-d6): δC
Activity assays in vitro
0.4 mM UDP-glucose was incubated with 0.2 mM aglycon, 50 178.3 (C-4), 161.4 (C-4′), 160.5 (C-7), 159.1 (C-5), 157.3 (C-2),
mM Tris-HCl (pH 7.4) and crude extracts of 20 mL strains 153.0 (C-9), 134.6 (C-3), 131.1 (C-3′′), 130.6 (C-2′/6′), 122.3 (C-
containing the recombinant EpGTs in a final volume of 100 μL. 1′), 122.1 (C-2′′), 114.1 (C-3′/5′), 108.3 (C-8), 105.6 (C-10), 102.0
Control experiments were performed with the crude extracts of (C-1′′′), 100.5 (C-1′′′′), 98.1 (C-6), 77.2 (C-5′′′′), 76.6 (C-3′′′′), 73.4
strains containing the empty vector. The activity assays, (C-2′′′′), 71.1 (C-4′′′), 70.7 (C-5′′′), 70.3 (C-3′′′), 70.1 (C-2′′′), 69.7
initiated by the addition of the enzymes, were performed at (C-4′′′′), 60.6 (C-6′′′′), 55.5 (OCH3-4′), 25.5 (C-5′′), 21.4 (C-1′′),
30 °C for up to 6 h and terminated by the addition of 200 μL of 17.9 (C-4′′), 17.5 (C-6′′′). ESI-MS: m/z 721 [M–H+COOH]–. All of
ice-cold MeOH. For activity assay in vitro of purified EpGT8 these data are in good agreement with the previously reported
(Ep7GT), EpGT2 and EpGT4, the reaction mixture containing 0.4 data for icariin11.
mM baohuoside (1), 0.8 mM UDPG, 5 μg of recombinant 7-O-β-D-[2-(N-acetylamino)-2-deoxy-glucopyranosyl]-
enzymes were incubated in a total volume of 100 μL at 30 °C baohuoside (1b, novel, isolated yield 19.7%).1H NMR (600 MHz,
and pH7.4 for 30 min. Subsequently, the samples were DMSO-d6): δH 7.86 (2H, d, J=8.9 Hz, H-2′/6′), 7.84 (1H, d, J=9.2
centrifuged at 15,000 g for 30 min to collect the supernatant Hz, H-NH), 7.11 (2H, d, J=8.9 Hz, H-3′/5′), 6.61 (1H,s, H-6), 5.27
and then analysed by HPLC-UV/ESIMS as described in the (1H,d, J=1.6 Hz, H-1′′′), 5.05 (1H, t, J=6.6 Hz, H-2′′), 5.02 (1H, d,
general methods. For quantification, three parallel assays were J=9.0 Hz, H-1′′′′), 3.85 (3H, s, O-CH3), 3.37 (2H, H-1′′), 3.04–3.98
routinely carried out. The HPLC peak areas were integrated with (protons in rhamnose and N-acetylglucosamine), 1.81 (3H, s, H-
Agilent Workstation Software, and the total percent conversion 8′′′′), 1.64 (3H, s, H-4′′), 1.59 (3H, s, H-5′′), 0.77 (3H, d, J=6.1 Hz,
was calculated as a percentage of the total peak areas of the H-6′′′); 13C NMR (125 MHz, DMSO-d6): δC 178.2 (C-4), 169.2 (C-
substrate and the product. The HPLC analyses were performed 7′′′′), 161.4 (C-4′), 160.4 (C-7), 159.1 (C-5), 157.5 (C-2), 153.0 (C-
on a Shiseido CAPCELL PAK C18 MG III column (250 mm × 4.6 9), 134.7 (C-3), 131.0 (C-3′′), 130.6 (C-2′/6′), 122.3 (C-1′), 122.2
mm I.D., 5 μm, Shiseido Co., Ltd., Tokyo, Japan) at a flow rate of (C-2′′), 114.0 (C-3′/5′), 108.4 (C-8), 105.7 (C-10), 102.0 (C-1′′′),
1 mL/min, with column temperature maintained at 30 °C. The 99.4 (C-1′′′′), 97.9 (C-6), 77.4 (C-5′′′′), 74.0 (C-3′′′′), 71.1 (C-4′′′),
mobile phase was a gradient elution of solvent A (0.1% formic 70.7 (C-5′′′), 70.3 (C-3′′′), 70.2 (C-2′′′), 70.1 (C-4′′′′), 60.6 (C-6′′′′),
acid aqueous solution) and solvent B (MeOH). Gradient 55.5 (OCH3-4′), 55.0 (C-2′′′′), 25.5 (C-4′′), 23.0 (C-8′′′′), 21.0 (C-
programmes were used for the analyses of the reactions (Table 1′′), 17.9 (C-5′′), 17.5 (C-6′′′). HRESIMS: m/z 762.2261 [M–
S1).
H+COOH]–.
7-O-β-D-Xylosyl-baohuoside (1c, novel, isolated yield 6.0%). 1H
NMR (600 MHz, DMSO-d6): δH 7.90 (2H, d, J=9.0 Hz, H-2′/6′),
7.13 (2H, d, J=9.0 Hz, H-3′/5′), 6.60 (1H, s, H-6), 5.28 (1H, d, J=1.7
Hz, H-1′′′), 5.16 (1H, t, J=6.6 Hz, H-2′′), 5.06 (1H, d, J=7.2 Hz, H-
1′′′′), 3.86 (3H, s, OCH3), 3.07–4.00 (protons in rhamnose and
xylose), 1.68 (3H, s, H-4′′), 1.60 (3H, s, H-4′′), 0.79 (3H, d, J=6.1
Hz, H-6′′′); 13C NMR (125 MHz, DMSO-d6): δC 178.3 (C-4), 161.3
(C-4′), 160.2 (C-7), 159.0 (C-5), 157.3 (C-2), 153.1 (C-9), 134.7 (C-
3), 131.1 (C-3′′), 130.5 (C-2′/6′), 122.3 (C-1′), 122.0 (C-2′′), 114.1
(C-3′/5′), 108.4 (C-8), 105.6 (C-10), 102.0 (C-1′′′), 100.7 (C-1′′′′),
97.9 (C-6), 77.2 (C-5′′′′), 76.3 (C-3′′′′), 73.1 (C-2′′′′), 71.1 (C-4′′′),
70.7 (C-5′′′), 70.3 (C-3′′′), 70.1 (C-2′′′), 69.3 (C-4′′′′), 55.5 (OCH3-
4′), 25.5 (C-5′′), 21.4 (C-1′′), 17.9 (C-4′′), 17.5 (C-6′′′). HRESIMS:
m/z 691.1915 [M–H+COOH]–.
Preparative scale reactions
Respectively, 19.5 μmol, 23.3 μmol and 23.3 μmol of
baohuoside (1), 14.0 μmol and 70.0 μmol of kaempferol (2)
were dissolved in 200 μL DMSO and diluted with a buffer
solution (50 mM Tris-HCl, pH 7.4, 25 mL total volume). UDP-
glucose, UDP-N-acetylglucosamine, UDP-xylose and UDP-
galacose were added to the same molar amount of aglycon 1
and twice the molar amount of aglycon 2, along with 25 mL of
the purified enzyme Ep7GT, respectively. The reactions were
performed at 30 °C for up to 12 h, followed by extraction with
ethyl acetate (100 mL × 5). The organic phase was evaporated
to dryness under reduced pressure, and the residue was
dissolved in 1.5 mL MeOH and purified by reverse-phase semi-
preparative HPLC. The obtained products were weighed, and
yields of 32.8% (1a, 6.4 μmol), 19.7% (1b, 4.6 μmol), 6.0% (1c,
1.4 μmol), 11.4% (2a, 8.0 μmol), 14.7% (2b, 10.3 μmol) and 17.1%
(2c, 2.4 μmol) were calculated, respectively. The structures
3,7,4′-Tri-O-β-D-glucosyl-kaempferol (2a, isolated yield 11.4%).
1H NMR (400 MHz, DMSO-d6): δH 8.14 (2H, d, J=9.0 Hz, H-2′/6′),
7.17 (2H, d, J=9.0 Hz, H-3′/5′), 6.82 (1H, d, J=2.2 Hz, H-8), 6.45
(1H, d, J=2.2 Hz, H-6), 5.49 (1H, d, J=7.1 Hz, H-1′′), 5.09 (1H, d,
J=7.2 Hz, H-1′′′), 5.03 (1H, d, J=7.3 Hz, H-1′′′′), 3.15–4.0 (protons
This journal is © The Royal Society of Chemistry 20xx
J. Name., 2013, 00, 1-3 | 7
Please do not adjust margins

 Feng, Keping
Feng, Keping
 Chen, Ridao
Chen, Ridao
 Xie, Kebo
Xie, Kebo
 Chen, Dawei
Chen, Dawei
 Liu, Jimei
Liu, Jimei
 Du, Wenyu
Du, Wenyu
 Yang, Lin
Yang, Lin
 Dai, Jungui
Dai, Jungui