1
044 J . Org. Chem., Vol. 64, No. 3, 1999
Notes
in THF (12.0 mL) and HMPA (3.0 mL) at -60 to -65 °C
internal) under Ar. After 2 min, CH I (2.5 mL, 40 mmol) was
added rapidly. The solution was warmed over 2 min to 10 °C,
quenched with saturated KH PO , and extracted with EtOAc.
The organic solution was dried (MgSO ), filtered, and concen-
trated. The residue (1.9 g) was purified by flash chromatography
80 g silica, 25% to 50% EtOAc-hexane) to give 0.62 g (82%) of
11â-Hyd r oxy-16r,17r,21-tr im eth ylp r egn a -1,4-d ien -3,20-
2
(
3
d ion e (Rim exolon e, 1). To a stirred, ice-cooled suspension of
3
9 (92 mg, 0.21 mmol) in 8 mL of CH OH under Ar was added
2
4
dropwise 0.40 mL of 6 M HCl. The mixture was allowed to warm
to room temperature over 1 h, and then 2.5 mL of THF was
added, whereupon the solid dissolved. After another 1.5 h, the
solution was diluted with water (100 mL), and the suspension
4
(
7
1
: mp 178-182 °C (dec); H NMR (CDCl
3
) δ 0.24 (s, 9H), 1.05
was extracted twice with CH
2 2
Cl . The combined organic extracts
(
t, 3H, J ) 7.3), 1.18 (s, 3H), 1.41 (s, 3H), 1.0-1.4 (m, 4H), 2.0-
were dried (MgSO ), filtered, and concentrated to give 88 mg of
4
2
6
.8 (m, 7H), 2.6 (q, 2H), 4.37 (br q, 1H, J ) 2.5), 5.99 (s, 1H),
.25 (dd, 1H, J ) 10, 2), 6.64 (q, 1H, J ) 2), 7.12 (d, 1H, J ) 10);
crude product, which was triturated twice with 5 mL of 2%
ether-hexane and dried under vacuum (Abderhalden, EtOH)
to give 77 mg (97%) of 1 as a white solid: mp 269-272 °C (dec),
1
3
3
C NMR (CDCl ) δ 0.97, 8.14, 18.18, 20.62, 30.28, 31.99, 32.00,
2
1
3
1
+
2.33, 33.70, 42.74, 44.17, 45.92, 56.80, 57.38, 71.43, 122.45,
28.08, 142.01, 155.13, 155.53, 169.92, 186.43, 199.44; [R]
lit. mp 230-273 °C (dec); H NMR (CDCl ) δ 0.90 (d, 3H, J )
3
2
3
D
7), 0.96 (s, 3H), 1.00 (s, 3H), 1.03 (t, 3H, J ) 7), 1.46 (s, 3H),
2.36 (q, 2H, J ) 7), 1.1-2.7 (m, 12H), 3.05 (m, 1H), 4.48 (br t,
1H, J ) 3), 6.01 (s, 1H), 6.27 (dd, 1H, J ) 10, 2), 7.30 (d, 1H, J
144° (c ) 0.05, CHCl
3
). Anal. Calcd for C25 Si: C, 72.76;
36 3
H O
H, 8.79. Found: C, 72.57; H, 8.73. Further elution provided 0.08
) 10); 13C NMR (CDCl ) δ 8.10, 14.93, 17.33, 18.44, 20.97, 31.54,
g (11%) of a 3:1 mixture of 5 and 7.
3
1
6r,21-Dim eth yl-11â,20-bis(tr im eth ylsiloxy)-p r egn a -1,4,-
32.01, 32.62, 32.87, 33.50, 33.90, 42.79, 44.09, 45.74, 50.19, 55.49,
63.47, 70.41, 122.36, 127.88, 156.10, 170.03, 186.53, 214.65;
1
7(20)-tr ien -3-on e (8). Methyllithium (1.0 M in 9:1 cumene-
THF, 4.0 mL, 4.0 mmol) was added dropwise via syringe over 5
min to a stirred, ice-cooled suspension of CuCN powder (187 mg,
[R]25 +94.1° (c ) 0.50, py), lit.
2
D
+100° (c ) 0.92, py). Anal. Calcd
for C24
H O ‚H O: C, 74.19; H, 9.34. Found: C, 74.08; H, 8.98.
34
3
2
2
.09 mmol) in THF (6.0 mL) under Ar. A yellow precipitate
formed and then dissolved during the addition of the first 2.0
mmol of CH Li. The resulting 0.2 M solution of (CH Cu(CN)-
Li
1
1â,17r-Dia cetoxyp r egn a -1,4-d ien -3,20-d ion e (11). p-Tol-
uenesulfonic acid monohydrate (0.03 g, 0.16 mmol) was added
to a stirred solution of TFAA (1.3 mL, 9.2 mmol) in HOAc (8
mL). The solution was heated at 37 °C for 5 min. Diol 3 (0.34 g,
3
3 2
)
2
was kept in an ice bath. To a stirred, cooled (-45 °C) solution
of 7 (0.395 g, 0.96 mmol) in 9.0 mL of THF under Ar was added
1
.0 mmol) was added, and heating (37 °C) and stirring were
continued for 5 h. The solution was poured cautiously into
saturated NaHCO (foaming, vigorous CO evolution), and the
TMSCl (0.50 mL, 4.0 mmol), followed by dropwise addition over
3
min of 5.0 mL (1.0 mmol) of the above THF solution of (CH
Cu(CN)Li . After 10 min, the mixture was quenched at -45 °C
by rapid addition of a solution of 0.5 mL (12 mmol) of CH OH
and 1.5 mL (11 mmol) of Et N and then warmed to 0 °C. Water
and EtOAc were added, and the solution was stirred vigorously.
The pH was adjusted from 9 to 7 with saturated KH PO , and
3 2
) -
3
2
2
product was isolated by EtOAc extraction. The EtOAc solution
was washed with water and brine, dried (MgSO4), filtered, and
concentrated. The crude product (0.39 g) was purified by
chromatography (50% EtOAc-hexane) to remove aromatized
material. The product was triturated with 1:1 ether-hexane to
3
3
2
4
stirring was continued for 0.5 h. The layers were separated, and
the organic solution was washed with water and brine, dried
1
afford 0.17 g (40%) of 11 as a white solid: mp 233-239 °C; H
NMR (DMSO-d ) δ 0.70 (s, 3H), 1.21 (s, 3H), 1.91 (s, 3H), 1.0-
6
(MgSO
4
), filtered, and concentrated. The residue was purified
2
.4 (m, 12H), 2.02 (s, 3H), 2.04 (s, 3H), 2.75 (dist t, 1H), 5.39 (br
by flash chromatography (40 g silica, 15% EtOAc-hexane) giving
s, 1H), 5.95 (s, 1H), 6.21 (dd, 1H, J ) 10, 2), 6.91 (d, 1H, J )
0
9
1
.41 g (85.5%) of 8 as a white solid: 1H NMR (CDCl
H), 0.24 (s, 9H), 0.96 (d, 3H, J ) 6.9), 1.01 (t, 3H, J ) 7.4),
.07 (s, 3H), 1.39 (s, 3H), 0.9-2.4 (m, 11H), 2.6 (br m, 3H), 4.38
3
) δ 0.20 (s,
1
3
9
2
0); 13C NMR (CDCl
) δ 16.61, 20.81, 21.23, 21.87, 24.07, 26.39,
3
0.19, 31.77, 31.92, 33.65, 36.42, 42.97, 46.02, 51.96, 53.77, 71.18,
6.08, 123.04, 128.59, 154.27, 168.32, 169.81, 170.42, 185.96,
(
)
br s, 1H), 6.01 (s, 1H), 6.28 (dd, 1H, J ) 10, 2), 7.12 (d, 1H, J
2
3
03.27. [R]
: C, 70.07; H, 7.53. Found: C, 69.88; H, 7.46.
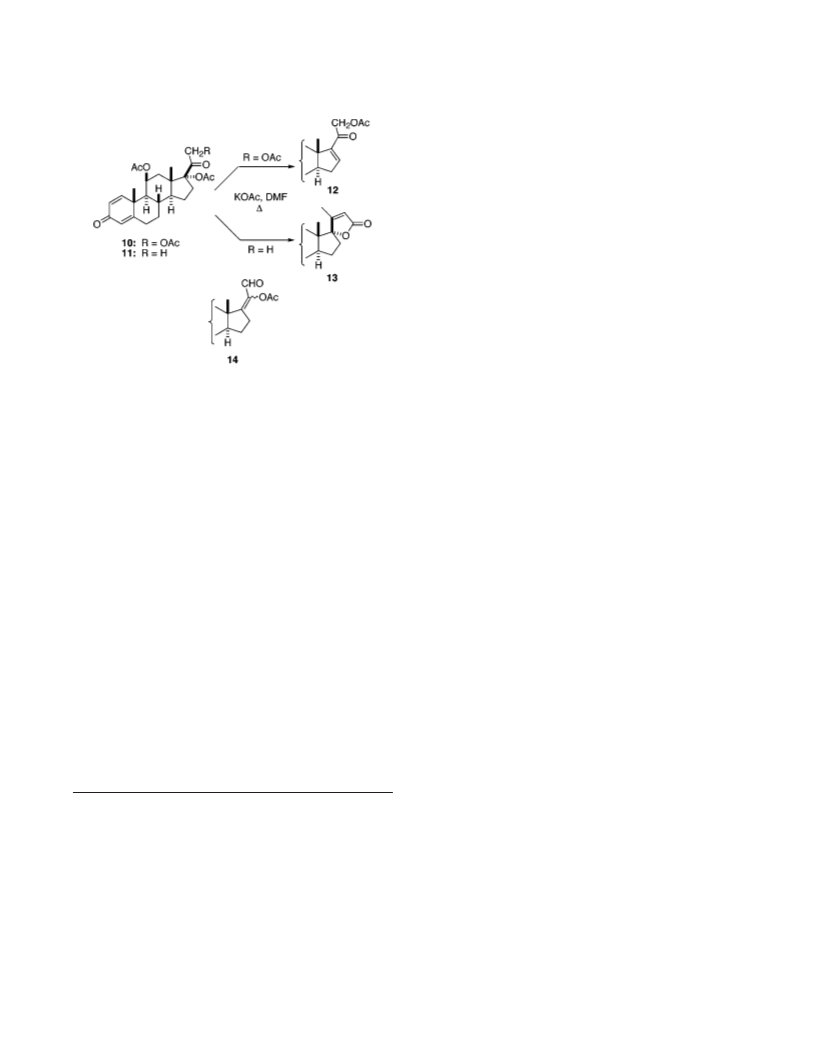
-(11â-Acet oxy-17r-h yd r oxy-3-oxo-1,4-p r egn a d ien -17â-
yl)-2-bu ten oic Acid γ-La cton e (13). A stirred solution of 11
68 mg, 0.16 mmol) and KOAc (0.15 g, 1.5 mmol) in anhydrous
D
+75.6° (c ) 0.39, CHCl
3
). Anal. Calcd for
2
3
10); [R]
D
3
+65.4° (c ) 0.51, CHCl ). A sample was recrystal-
25 32 6
C H O
lized from diisopropyl ether giving an E/Z mixture (NMR): mp
3
1
94-206 °C. Anal. Calcd for C29
Found: C, 69.66; H, 9.73.
1â-(Tr im et h ylsiloxy)-16r,17r,21-t r im et h ylp r egn a -1,4-
48 3 2
H O Si : C, 69.54; H, 9.66.
(
1
1
DMF (1.2 mL) was heated under Ar to 105 °C for 1.5 h and then
to 120 °C for 1 h. TLC showed only starting 11. KOAc (0.2 g, 2
mmol) was added, and heating was continued for 15 h, during
which time the temperature rose to 143 °C. The cooled mixture
was partitioned between EtOAc and water; the organic solution
d ien -3,20-d ion e (9). Molecular sieves (4A, /16 in. spheres, 2.7
1
9
g, dried at 250 °C) and dry benzyltrimethylammonium fluoride
0.8 g) were weighed under Ar (glovebag) into an oven-dried Ar-
(
flushed 25-mL flask containing a stir bar. THF (3.0 mL) was
added via syringe, and the mixture was stirred rapidly for 6 h.
To the resulting paste was added via syringe a solution of 8 (47
mg, 0.094 mmol) in CH I (1.3 mL, 21 mmol, dried over 4A
3
molecular sieves). After 45 min of stirring at room temperature,
EtOAc was added and the suspension was filtered. The filtrate
4
was dried (MgSO ), filtered, and concentrated; and the residue
was purified by flash chromatography on silica (50% EtOAc-
hexanes) to afford 17.5 mg (26%) of 11, followed by 31 mg (47.5%)
1
of 13 as an oil: H NMR (CDCl
3
) δ 1.11 (s, 3H), 1.28 (s, 3H),
1
1
6
.1-2.7 (m, 12H), 2.10 (s, 3H), 2.13 (d, 3H, J ) 1.5), 5.39 (br s,
H), 5.50 (br q, 1H, J ) 3), 5.81 (d, 1H, J ) 1.5), 6.04 (s, 1H),
.27 (dd, 1H, J ) 10, 2), 6.90 (d, 1H, J ) 10); 13C NMR (CDCl
4
was washed with half-saturated brine, dried (MgSO ), eluted
through Florisil with EtOAc, and concentrated to give 46.5 mg
of an oil that solidified. Crystallization from 12% EtOAc-
hexanes (6 mL) at -25 °C followed by drying under vacuum at
3
)
δ 16.47, 18.41, 20.85, 21.83, 24.31, 31.66, 31.84, 32.34, 33.40,
4.88, 42.83, 48.96, 51.57, 53.46, 71.21, 99.33, 120.15, 123.00,
28.49, 154.05, 168.00, 168.05, 169.62, 170.92, 185.89; HRMS
3
1
7
5 °C afforded 26 mg (63%) of 9 as a white solid. A chromato-
graphically purified sample of 9 showed the following charac-
1
30 5
Calcd for C25H O 410.20932, found 410.20837.
teristics: mp 207.5-210 °C; H NMR (CDCl
3
) δ 0.20 (s, 9H), 0.87
(
d, 3H, J ) 7.2); 0.94 (s, 6H), 1.04 (t, 3H, J ) 7.1), 1.37 (s, 3H);
2
1
)
.34 (q, 2H, J ) 7), 1.1-2.7 (m, 11H), 3.07 (m, 1H), 4.46 (br t,
Su p p or tin g In for m a tion Ava ila ble: Infrared and mass
spectra (2 pages). This material is contained in libraries on
microfiche, immediately follows this article in the microfilm
version of the journal, and can be ordered from the ACS; see
any current masthead page for ordering information.
H, J ) 3), 6.00 (s, 1H), 6.27 (dd, 1H, J ) 10, 2), 7.10 (d, 1H, J
1
3
3
10); C NMR (CDCl ) δ 0.95, 8.17, 14.59, 17.24, 18.14, 20.45,
3
5
2
1.58, 32.06, 32.42, 32.65, 33.38, 33.94, 40.90, 43.90, 45.77, 50.68,
6.29, 63.67, 71.14, 122.35, 128.03, 155.38, 170.14, 186.33,
2
3
14.20; [R]
D
3 42 3
+64.6° (c ) 0.50, CHCl ). Anal. Calcd for C27H O -
Si: C, 73.25; H, 9.56. Found: C, 73.00; H, 9.65.
J O981848X

 Conrow, Raymond E.
Conrow, Raymond E.