Organic Process Research & Development
Communication
column chromatography (hexane/ethyl acetate = 100/0 to 0/
Hz, 1H), 8.09 (dd, J = 8.8 Hz, 2.4 Hz, 1H), 7.00 (d, J = 8.8 Hz,
1H), 4.36 (q, J = 7.2 Hz, 2H), 3.90 (d, J = 6.4 Hz, 2H), 2.77 (s,
3H), 2.23−2.19 (m, 1H), 1.39 (t, J = 7.2 Hz, 3H), 1.09 (d, J =
6.8 Hz, 6H).
1
00) to yield 4a (611 mg from the solid and 405 mg from the
filtrate; 91% yield in total) as an off-white solid. The NMR
4
spectrum was consistent with that reported previously.
Second-Generation C−H Arylation: Reaction Screen-
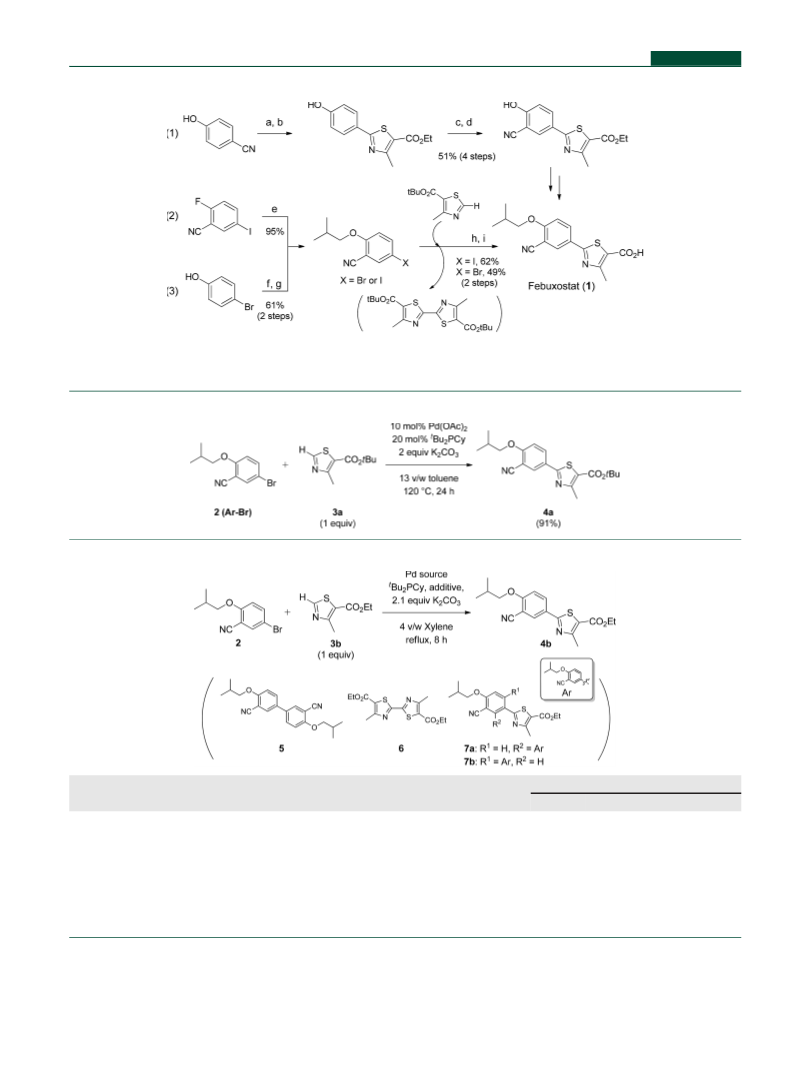
ing (Table 1). Ethyl 2-(3-Cyano-4-isobutoxyphenyl)-4-
methylthiazole-5-carboxylate (4b). To a three-neck flask,
2-(3-Cyano-4-isobutoxyphenyl)-4-methylthiazole-5-
carboxylic Acid (Febuxostat, 1). To a three-neck flask were
added 4b (25.0 g, 72.6 mmol), THF (25 mL), and ethanol
(175 mL). The mixture was warmed to 40−50 °C, and 8 N
aqueous NaOH (10.9 mL, 87.1 mmol) was added to the
mixture. The mixture was stirred at the same temperature until
the conversion reached 99.5%. Water (76.2 mL) was added to
the mixture, and the resulting mixture was stirred for 1 h at the
same temperature, followed by quenching with 1 M aqueous
HCl (95.8 mL, 95.8 mmol) to adjust the pH to below 3.5. To
the mixture was added water (62.5 mL) at 60−70 °C, and the
resulting mixture was then heated to the reflux temperature,
stirred for 30 min, and cooled to 30−40 °C. The resulting
slurry was filtered, and the wet cake was washed with warm
3
b (1.71 g, 10.0 mmol), 2 (2.54 g, 10.0 mmol), palladium
catalyst, di-tert-butyl(cyclohexyl)phosphine, additive, K CO
2
3
(
2.90 g, 21.0 mmol), and xylene (10 mL) were added under
2
N . The mixture was stirred at 140−145 °C and periodically
monitored by HPLC.
C−H Arylation: Role of the Reagents (Table 2). Ethyl
-(3-Cyano-4-isobutoxyphenyl)-4-methylthiazole-5-carbox-
2
ylate (4b). N was vigorously flowed into a sealed tube, to
2
which 3b (2.14 g, 12.5 mmol), 2 (2.54 g, 10.0 mmol), PdCl2
(
(
4.4 mg, 0.025 mmol), di-tert-butyl(cyclohexyl)phosphine
22.8 mg, 0.10 mmol), isobutyric acid (8.8 mg, 0.10 mmol),
copper(I) bromide−dimethyl sulfide complex (206 mg, 1.00
mmol), K CO (2.90 g, 21.0 mmol), and/or xylene (7.5 mL)
water (625 mL) and dried in vacuo at 75−85 °C to give 1
1
(22.5 g, 98%) as a whitish solid. H NMR (400 MHz, CDCl
):
3
2
3
were added. The tube was quickly closed, and the mixture was
δ 8.20 (d, J = 2.4 Hz, 1H), 8.11 (dd, J = 9.0 Hz, 2.4 Hz, 1H),
7.03 (d, J = 9.0 Hz, 1H), 3.91 (d, J = 6.6 Hz, 2H), 2.80 (s,
3H), 2.23−2.20 (m, 1H), 1.20 (d, J = 6.8 Hz, 6H).
stirred in an aluminum block at 140 °C for 24 h. CAUTION!
The internal pressure greatly increases because of CO and water
2
evolution! The mixture was diluted and measured by HPLC.
Optimized C−H Arylation: Ethyl 2-(3-Cyano-4-iso-
butoxyphenyl)-4-methylthiazole-5-carboxylate (4b). To
a mixture of potassium tert-butoxide (11.8 g, 105 mmol) and
toluene (100 mL) was added 2-methyl-1-propanol (8.15 g, 110
mmol) dropwise. The mixture was stirred for 5 min, and
approximately two-thirds of the mixture removed at 60 °C
AUTHOR INFORMATION
■
*
ORCID
Notes
under reduced pressure. N was then back-filled into the vessel,
2
and the mixture was stirred in an ice bath. To the resulting
mixture of potassium isobutoxide was added 8 (20.0 g, 100
mmol) in toluene (50 mL) dropwise as the internal
temperature was maintained between 5 and 10 °C. The
mixture was warmed to room temperature and stirred for 2 h.
After reaction completion, the mixture was washed sequentially
with 20 wt % citric acid (50 mL) and water (50 mL, twice).
The resulting organic layer was concentrated at 60 °C under
reduced pressure to give 71.6 g of 2 (53 mL of toluene was
theoretically assayed).
The authors declare no competing financial interest.
ACKNOWLEDGMENTS
■
We thank Ms. Miki Murata and Mr. Takahiro Takeuchi for
analytical support and Mr. Tomohide Ida, Dr. Tomohiro
Shimada, Dr. Shiro Kondo, and Mr. Akira Nakano for helpful
discussions.
REFERENCES
■
To the solution of 2 in a three-neck flask with a Dean−Stark
(
1) (a) Osada, Y.; Tsuchimoto, M.; Fukushima, H.; Takahashi, K.;
1
6
trap were added 3b (18.0 g, 105 mmol), PdCl (18 mg, 0.10
Kondo, S.; Hasegawa, M.; Komoriya, K. Hypouricemic Effect of the
Novel Xanthine Oxidase Inhibitor, TEI-6720, in Rodents. Eur. J.
Pharmacol. 1993, 241, 183. (b) Komoriya, K.; Osada, Y.; Hasegawa,
M.; Horiuchi, H.; Kondo, S.; Couch, R. C.; Griffin, T. B.
Hypouricemic Effect of Allopurinol and the Novel Xanthine Oxidase
Inhibitor TEI-6720 in Chimpanzees. Eur. J. Pharmacol. 1993, 250,
2
mmol), di-tert-butyl(cyclohexyl)phosphine (55 mg, 0.24
mmol), isobutyric acid (35 mg, 0.40 mmol), copper(I)
bromide (143 mg, 1.00 mmol), KHCO (21.0 g, 210 mmol),
3
and toluene (23 mL, in addition to the aforementioned 53
mL) under N . After 5 min of stirring at room temperature, the
2
4
55. (c) Horiuchi, H.; Ota, M.; Kobayashi, M.; Kaneko, H.; Kasahara,
mixture was warmed and stirred at the reflux temperature for
Y.; Nishimura, S.; Kondo, S.; Komoriya, K. A Comparative Study on
the Hypouricemic Activity and Potency in Renal Xanthine Calculus
Formation of Two Xanthine Oxidase/Xanthine Dehydrogenase
Inhibitors: TEI-6720 and Allopurinol in Rats. Res. Commun. Mol.
Pathol. Pharmacol. 1999, 104, 307. (d) Okamoto, K.; Eger, B. T.;
Nishino, T.; Kondo, S.; Pai, E. F.; Nishino, T. An Extremely Potent
Inhibitor of Xanthine Oxidoreductase. J. Biol. Chem. 2003, 278, 1848.
1
5 h. After reaction completion, the mixture was cooled to an
internal temperature of 90 °C. The insoluble species were then
filtered off (remaining at this temperature) and washed with
hot toluene (25 mL). The filtrate was then partially
concentrated at 60 °C under reduced pressure. To the
concentrated mixture (53 mL of toluene was assayed),
additional toluene (23 mL) and 90% aqueous EtOH (178
mL) were added. The mixture was dissolved at 85 °C and then
cooled to 60−68 °C, inducing precipitation. The mixture was
stirred at the same temperature for 1 h, cooled to 0 °C over 2
h, and stirred at 0 °C for 1.5 h. The resulting slurry was filtered,
and the wet cake was washed with 80% aqueous EtOH (76
(
e) Takano, Y.; Hase-Aoki, K.; Horiuchi, H.; Zhao, L.; Kasahara, Y.;
Kondo, S.; Becker, M. A. Selectivity of Febuxostat, a Novel Non-
purine Inhibitor of Xanthine Oxidase/Xanthine Dehydrogenase. Life
Sci. 2005, 76, 1835.
(2) (a) Hande, K. R.; Noone, R. M.; Stone, W. J. Severe Allopurinol
Toxicity. Description and Guidelines for Prevention in Patients with
Renal Insufficiency. Am. J. Med. 1984, 76, 47. (b) Mayer, M. D.;
Khosravan, R.; Vernillet, L.; Wu, J.-T.; Joseph-Ridge, N.; Mulford, D.
J. Pharmacokinetics and Pharmacodynamics of Febuxostat, a New
mL) and dried in vacuo at 80 °C to give 4b (31.3 g, 91%) as a
1
whitish solid. H NMR (400 MHz, CDCl ): δ 8.18 (d, J = 2.4
3
E
Org. Process Res. Dev. XXXX, XXX, XXX−XXX

 Dohi, Masahiko
Dohi, Masahiko
 Kato, Yoshiaki
Kato, Yoshiaki
 Komiyama, Masato
Komiyama, Masato
 Kurokawa, Masayuki
Kurokawa, Masayuki
 Minamizono, Kunio
Minamizono, Kunio
 Sato, Yoshinori
Sato, Yoshinori
 Teramoto, Mitsuru
Teramoto, Mitsuru
 Tsuchiya, Hideyoshi
Tsuchiya, Hideyoshi
 Tsuchiya, Naoki
Tsuchiya, Naoki
 Yajima, Naoki
Yajima, Naoki