3
34
M.N. Timofeeva et al. / Journal of Molecular Catalysis A: Chemical 363–364 (2012) 328–334
at 18% conversion of ␣-pinene for 5 h. The conversion of ␣-pinene
reduces with decrease in catalyst amount (Table 5).
for productive and beneficial discussion of our results. The authors
would like to thank Dr. S.A. Prihod’ko for GC-MASS measurements
and discussion.
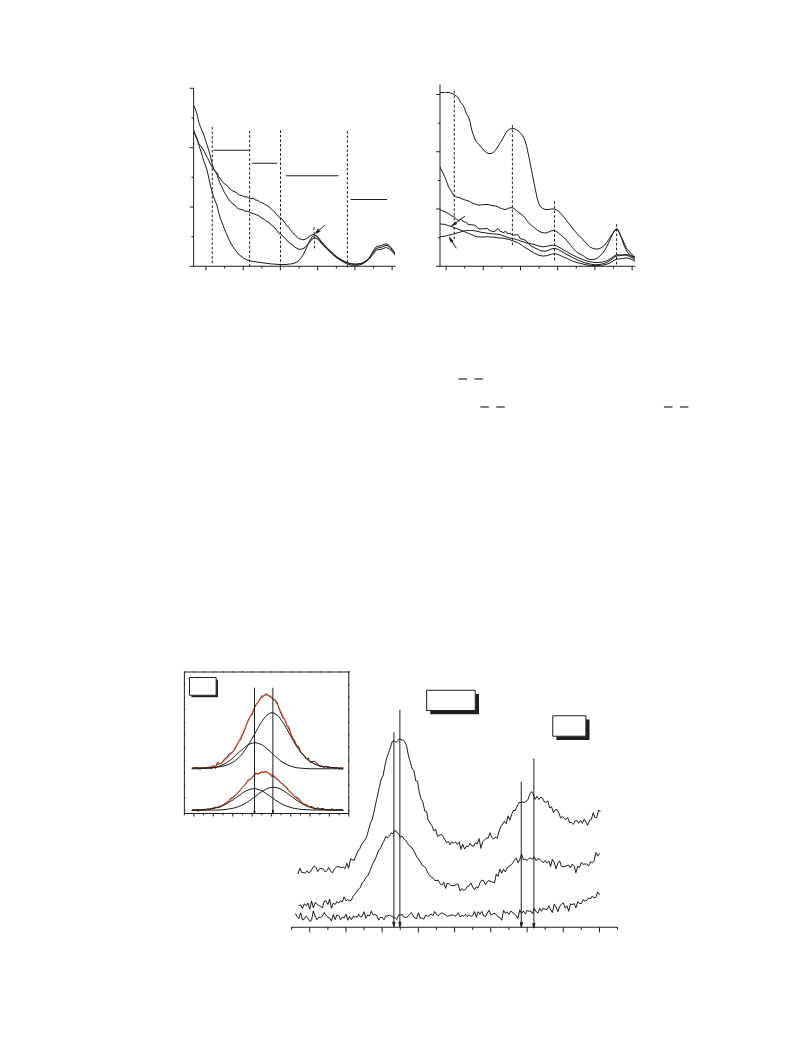
The catalytic behavior of V-VSB-5 may be related to the presence
5
+
of tetrahedral V species and their one-electron transfer processes,
i.e. V5 /V valence changes. To obtain information on the oxida-
tion state of vanadium in situ UV–vis spectra of 1.7% V-VSB-5 in the
presence of ␣-pinene were obtained (Fig. 3B). As can be seen from
Fig. 3B (curves 1 and 2) spectrum of ␣-pinene adsorbed on 1.7% V-
+
4+
References
[1] H.O. Pastore, S. Coluccia, L. Marchese, Annu. Rev. Mater. Res. 35 (2005) 351.
[2] M. Hartmann, L. Kevan, Chem. Rev. 99 (1999) 635.
[3] J.-S. Chang, S.-E. Park, Q. Gao, G. Ferey, A.K. Cheetham, Chem. Mater. 9 (2001)
859.
◦
VSB-5 in N2 atmosphere at 25 C is similar to the spectrum of 1.7%
◦
[4] C. Li, H. Kawada, X. Sun, H. Xu, Y. Yoneyama, N. Tsubaki, ChemCatChem. 3 (2011)
84.
V-VSB-5. After heating at 70 C for 30 min and then adsorption of
6
␣
-pinene on 1.7% V-VSB-5 in N atmosphere, two strong bands at
2
[
5] S.H. Jhung, J.-H. Lee, A.K. Cheetham, G. Ferey, J.-S. Chang, J. Catal. 239 (2006)
97.
−1
3
3
V
1,450 and ∼40,000 cm are observed in spectrum (Fig. 3B, curve
), which can be evidently attributed to the LMCT of T -coordinated
[6] N. Guillou, Q. Gao, M. Nogues, R. Morris, M. Hervieu, G. Ferey, A.K. Cheetham,
C. R. Acad. Sci. Ser. IIc 2 (1999) 387.
d
5
+
4+
and V ions [45]. The intensities of these bands decrease after
[
7] M.N. Timofeeva, Z. Hasan, A. Yu Orlov, V.N. Panchenko, Yu A. Chesalov, I.E.
Soshnikov, S.H. Jhung, Appl. Catal. B: Environ. 107 (2011) 197.
◦
heating in air at 70 C (Fig. 3B, curve 4). Note that intensity of band
−1
at ∼40,000 cm changes largely in comparison with that of band at
[8] S.-J. Liu, H.-Y. Cheng, F.-Y. Zhao, J.-Y. Gong, S.-H. Yu, Chem. Eur. J. 14 (2008)
4074.
−1
3
1,450 cm . The obtained results can indicate that vanadium ions
[
9] D. Gao, Q. Gao, Catal. Commun. 8 (2007) 681–685.
are active sites for adsorption of ␣-pinene molecules and following
[
10] J. Yu, A. Wang, J. Tan, X. Li, J.A. van Bokhoven, Y. Hu, J. Mater. Chem. 18 (2008)
3601.
its catalytic oxidation. These data are in agreement with a cor-
relation between conversion of ␣-pinene and V5 /V molar ratio
+
4+
[11] S.H. Jhung, J.-S. Chang, Y.K. Hwang, J.-M. Greneche, G. Ferey, A.K. Cheetham, J.
Phys. Chem. B 109 (2005) 845.
5
+
4+
(
Fig. 6B). Of note that similar correlation between V /V molar
[
12] S.H. Jhung, J.-S. Chang, J.W. Yoon, J.-M. Greneche, G. Ferey, A.K. Cheetham, Chem.
Mater. 16 (2004) 5552.
5+
ratio and catalytic activity, i.e. the importance of V centers, was
demonstrated in the selective oxidation of butane to maleic anhy-
dride over vanadium phosphorus catalysts [46,47].
[
13] G. Centi, S. Perathoner, F. Trifiro, A. Aboukais, C.F. Aissi, M. Guelton, J. Phys.
Chem. 96 (1992) 2617.
[14] G. Centi, F. Trifiro, Appl. Catal. A: Gen. 143 (1996) 3.
[15] T. Blasco, J.M. Lopez Nieto, A. Dejoz, M.I. Vazquez, J. Catal. 157 (1995) 271.
[16] S.J. Kulkarni, R. Rao, M. Subrahmanyam, A.V.R. Rao, A. Sarkany, L. Guczi, Appl.
Catal. A: Gen. 139 (1996) 59.
4
. Conclusion
[
17] W.F. Erman, Chemistry of the Monoterpenes: An Encyclopedic Handbook,
Dekker, New York, 1985.
In this study physicochemical properties of vanadium-
containing nickel phosphate molecular sieves (V-VSB-5) were
[
[
18] P.A. Wende, T.P. Muciaro, J. Am. Chem. Soc. 114 (1992) 5878.
19] T. Joseph, D.P. Sawant, C.S. Gopinath, S.B. Halligudi, J. Mol. Catal. A: Chem. 184
investigated by XRD, SEM, XPS, N -adsorption/desorption anal-
2
(
2002) 289.
ysis DR-UV–vis and FT-IR spectroscopy. According to XPS and
[20] E. Blaz, J. Pielichowski, Molecules 11 (2006) 115.
[21] M.V. Patil, M.K. Yadav, R.V. Jasra, J. Mol. Catal. A 277 (2007) 72.
DR-UV–vis spectroscopy both V4 and V states are observed in
+
5+
[22] C.D. Pina, E.F. Falletta, M. Rossi, Chem. Soc. Rev. 41 (2012) 350.
[23] P. Selvam, V.M. Ravat, V. Krishna, J. Phys. Chem. C 115 (2011) 1922.
[24] R. Raja, G. Sankar, J.M. Thomas, Chem. Commun. (1999) 829.
V-VSB-5 samples. The increase in vanadium content leads to the
5
+
4+
increase V /V molar ratio. It was shown by FT-IR spectroscopy
using CDCl3 as a probe molecule that the strength of basic sites for
[25] W.J. Yang, C.C. Gui, Z.Y. Li, N.Y. Tao, J. Porphyr. Phthalocyan. 13 (2009) 973.
[26] S. Bhattachacharjee, T.J. Dines, J.A. Anderson, J. Catal. 225 (2004) 398.
[
−1
−1
V-VSB-5 (843 kJ mol ) is close to that for VSB-5 (843 kJ mol ). The
Brönsted acidity was investigated by adsorption of two indicators,
such as pyridine and phenolic red. It was found that the strength
of Brönsted acid sites increased with increasing of vanadium con-
tent in V-VSB. The increase in vanadium content leads to increase
in molar ratio of Brönsted/Lewis acid sites.
27] J.E. Ancel, N.V. Maksimchuk, I.L. Simakova, V.A. Semikolenov, Appl. Catal. A:
Gen. 272 (2004) 109.
[28] Y.-W. Suh, N.-K. Kim, W.-S. Ahn, H.-K. Rhee, J. Mol. Catal. A: Chem. 174 (2001)
49.
2
[
29] P.A. Robles-Dutenhefner, M.J. Silva, L.S. Sales, E.M.B. Sousa, E.V. Gusevskaya, J.
Mol. Catal. A: Chem. 217 (2004) 139.
[30] J.H. Scofield, J. Electron Spectrosc. Relat. Phenom. 8 (1976) 129.
[
31] A.A. Davydov, Molecular Spectroscopy of Oxide Catalyst Surfaces, John Wiley,
England, 2003, p. 1.
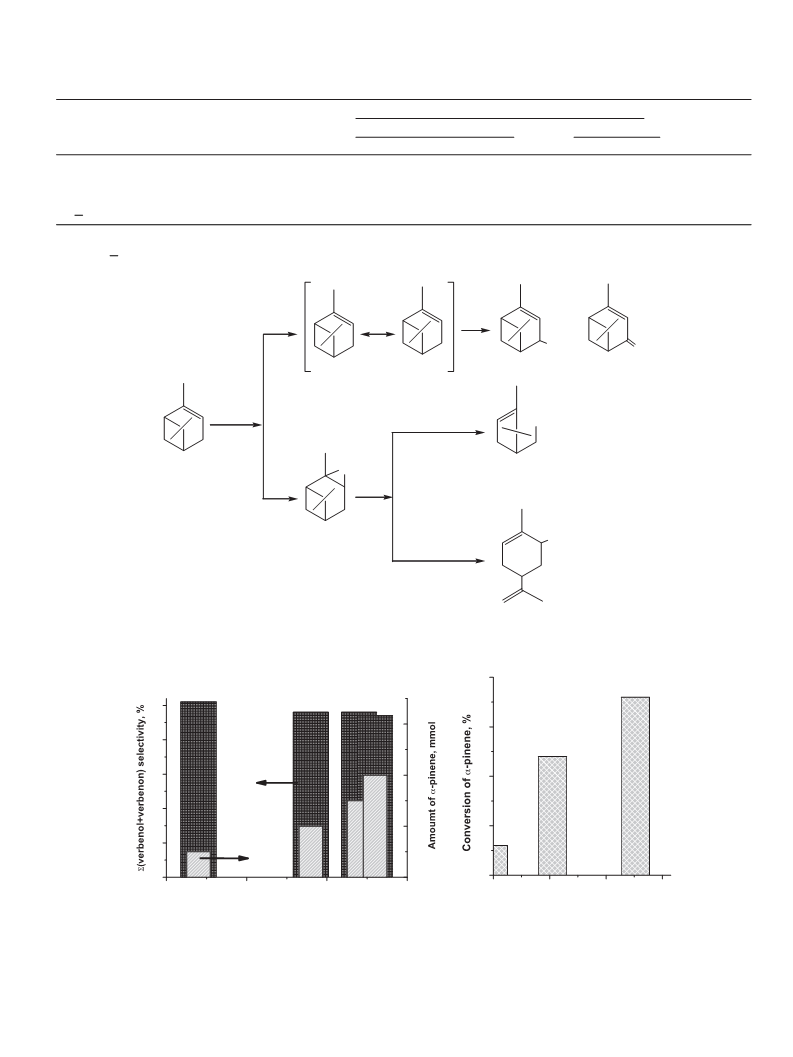
The catalytic activity of V-VSB-5 was tested for the oxidation
of ␣-pinene with molecular oxygen. The main reaction products
are ␣-pinene epoxide, ␣-campholene aldehyde, trans-carveol, ver-
benol and verbenone. It was shown that conversion of ␣-pinene
and distribution of products depends on the vanadium content
in V-VSB-5 materials. As the vanadium content in VSB-5 network
increased, the conversion of ␣-pinene increased. Acid–base proper-
ties of V-VSB-5 affect the distribution of by-products formed via the
isomerization of ␣-pinene oxide over Lewis acid sites and Brönsted
acid sites to campholenic aldehyde and trans-carveol, respectively.
The increase in vanadium content in V-VSB-5 leads to the increase
in isomerization of ␣-pinene oxide to trans-carveol and decrease
in that to campholenic aldehyde. From DR-UV–vis studies we con-
clude that the active center for ␣-pinene constitute a V5 ions that
is in agreement with a correlation between conversion of ␣-pinene
and V5+/V molar ratio.
[
32] F.A. Cotton, G. Wilkison, Advanced Inorganic Chemistry, Wiley, New York, 1980,
p. 786.
[33] X. Gao, M.A. Banares, I.E. Wachs, J. Catal. 188 (1999) 325.
[34] A. Corma, J.M. Lopez Nieto, N. Paredes, Appl. Catal. A: Gen. 104 (1993) 161.
[35] T. Sen, V. Ramaswamy, S. Ganapathy, P.R. Rajamohanan, S. Sivasanker, J. Phys.
Chem. 100 (1996) 3809.
[36] A. Albuquerque, H.O. Pastore, L. Marchese, Stud. Surf. Sci. Catal. 158 (2005) 901.
[
37] M. Vishnuvarthan, A.J. Paterson, R. Raja, A. Piovano, F. Bonino, E. Gianotti, G.
Berlier, Micropor. Mesopor. Mater. 138 (2011) 167.
[
38] M.H. Zahedi-Niaki, S.M.J. Zaidi, S. Kaliaguine, Appl. Catal. A: Gen. 196 (2000)
9.
[39] M. Demeter a, M. Neumann, W. Reichelt, Surf. Sci. 454–456 (2000) 41.
[40] G.A. Sawatzky, D. Post, Phys. Rev. B 20 (1979) 1546.
[41] M.J. Haanepen, J.H.C. van Hooff, Appl. Catal. A: Gen. 152 (1997) 183.
[42] R.A. Sheldon, J.K. Kochi, Metal-Catalyzed Oxidations of Organic Compounds,
Academic Press, London, 1981.
+
[
43] G. Neri, G. Rizzo, C. Crisafulli, L. De Luca, A. Donato, M.G. Musolino, R.
Pietropaolo, Appl. Catal. A: Gen. 295 (2005) 116.
4+
[44] W.F. Holderich, J. Roseler, G. Heitmann, A.T. Liebens, Catal. Today 37 (1997)
53.
45] R. Bulanek, L. Capek, M. Setnicka, P. Cicmanec, J. Phys. Chem. C 115 (2011)
2430.
3
[
[
[
Acknowledgements
1
46] G.J. Hutchings, C.J. Kiely, M.T. Sananes-Schulz, A. Burrows, J.C. Volta, Catal.
Today 40 (1998) 273.
This work was partly supported by Kyungpook National Uni-
versity Research Fund, 2011. We also thank Dr. N.V. Maksimchuk
47] M. Abon, J.C. Volta, Appl. Catal. A: Gen. 157 (1997) 173.

 Timofeeva, M. N.
Timofeeva, M. N.
 Panchenko, V. N.
Panchenko, V. N.
 Prosvirin, I. P.
Prosvirin, I. P.
 Hasan, Zubair
Hasan, Zubair
 Jhung, Sung Hwa
Jhung, Sung Hwa