Enantioselective Enzymatic Cleavage of N-Benzyloxycarbonyl Groups
FULL PAPERS
for 30 min to obtain a cell extract. Protein in cell extracts was
estimated by Bio-Rad protein reagent using bovine serum
albumin as a standard. The assay mixture contained 1 ± 10 mL
of enzyme fraction, 0.8 mL water and 0.2 mL Bio-Rad reagent.
After mixing, the absorbance of the solution was measured at
The column was equilibrated with 12.5% methanol in 3 mM
CuSO . The run time was 30 min with 0.8 mL/min flow rate.
4
The detector was set at 240 nm. Retention times were: d-
phenylalanine, 8.25 min; l-phenylalanine, 9.63 min; d-tyro-
sine, 5.54 min; l-tyrosine, 4.89 min; d-proline, 2.32 min; l-
proline, 2.54 min.
595 nm.
Biotransformation using Cell Extract
Purification of Cbz-Deprotecting Enzyme
Various Cbz-protected amino acids (10 mg) and other sub-
strates were incubated with the cell extracts (10 mL) at 428C
for 18 ± 20 h. The reactions were terminated by addition of 2
volumes of 50% acetonitrile containing 0.4% trifluoroacetic
acid (TFA). The reaction mixtures were analyzed for substrate
and product concentration by HPLC.
All the purification steps were carried out at room temperature
using Cbz-l-phenylalanine as a substrate. Typical assay mix-
ture contained 0.4 mL enzyme (cell extracts or fractions) and
0.5 mg of Cbz-l-phenylalanine. The reaction was carried out at
458C for 4 h. The reaction mixture was quenched with 0.4 mL
of 50% acetonitrile containing 0.4% trifluoroacetic acid. The
samples were filtered and analyzed by HPLC. Preparation of
cell extract from 100 g of washed cells (S. paucimobilis SC
1
6113) was carried out as described above. The cell extract was
HPLC Analysis
loaded onto a Whatman DE-52 column (200 mL packed bed)
equilibrated with buffer A. The column was washed with
2
Analysis was performed using a Hewlett-Packard (HP) 1090
instrument with a Vydac C-18 reverse phase column. The
mobile phase consisted of solvent A containing 0.1% trifluoro-
acetic acid (TFA) in water and solvent B containing 0.1% TFA
in 70% acetonitrile:30% water. The following gradient of
solvent A and B was used for the separation of substrates and
products. 0 min: A 100%, 0 ± 15 min: A 50% and B 50%, 15 ±
00 mL of buffer A and eluted with a 1-L gradient of buffer A
containing NaCl from 0.2 ± 0.6 M. Fractions of 20 mL were
collected. Enzyme does not bind to DE-52 column and activ-
ity was eluted in the wash buffer. The pooled wash from the
DE-52 column containing the enzyme activity was precipitated
with ammonium sulfate (516 g/L ammonium sulfate added).
The resulting precipitated protein was collected by centrifu-
gation at 15,000 rpm for 20 min at 48C, dissolved in buffer A
containing 1 M ammonium sulfate and loaded on to a
25 min: B 100%, 25 ± 26 min: A 100%, and 26 30 min: A 100%.
The flow rate was 1 mL/min. The column temperature was
ambient, and the detection wavelength was 215 nm. Under
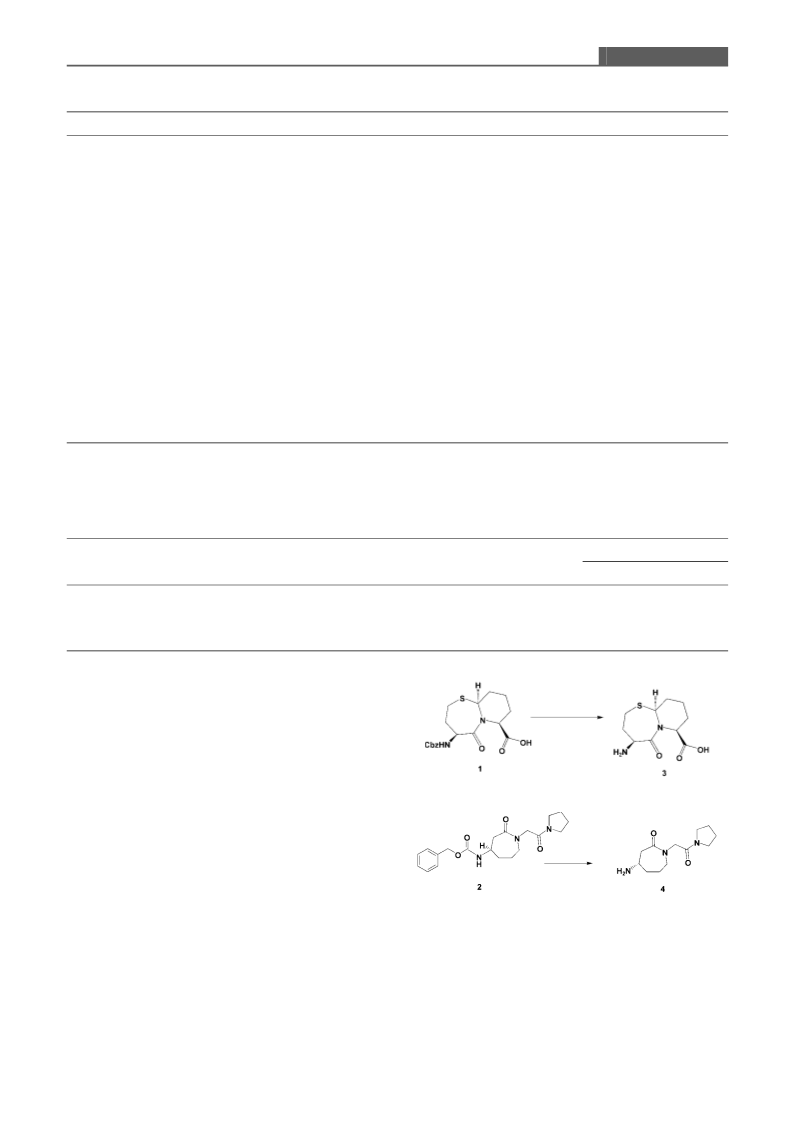
these conditions, the retention times for [4S-(4a,7a,10ab)ꢀ-
octahydro-5-oxo-4-{[(phenylmethoxy)carbonylꢀ-amino}-7H-
pyrido[2,1-bꢀ[1,3ꢀthiazepine-7-carboxylic acid 1 and its depro-
tected analogue 3 were 15.48 min and 28 min, respectively. The
retention times for N-a-Cbz-l-phenylalanine and l-phenyl-
alanine were 16.99 min and 7.35 min, respectively. All other
Cbz-containing compounds were analyzed using the above
HPLC method.
Analyses of the [(3S)-hexahydro-2-oxo-1-[2-oxo-2-(1-pyr-
rolidinyl) ethylꢀ-1H-azepin-3-ylꢀcarbamic acid phenylmethyl
ester 2 and its deprotected analogue 4 were carried out on a
YMC basic column (5 mm; 4.6 Â 150 mm). The mobile phase
consisted of solvent A containing 0.1% H PO in water and
¾
Pharmacia fast flow Phenyl Sepharose column (50 mL bed
volume) equilibrated with buffer A containing 132 g/L ammo-
nium sulfate (1 M ammonium sulfate). The column was
washed with 75 mL of buffer A containing 1 M ammonium
sulfate and then with buffer A containing 0.5 M and 0.25 M
ammonium sulfate and finally with buffer A without any
ammonium sulfate. Fractions of 5 mL were collected. The most
active fractions (fractions eluted from buffer A without
ammonium sulfate) were pooled and concentrated by ultra-
filtration to 8 mL using an Amicon YM -10 membrane. The
concentrated material was loaded on to a Sephacryl S-200¾
column (400 mL) equilibrated with buffer A. The protein was
eluted with buffer A at a flow rate of 0.8 mL/min. Fractions of
3
4
1
0 mL were collected. Active fractions containing enzyme
solvent B containing 90% acetonitrile in water. The linear
gradient of solvent A and B over 20 min was used for the
separation of substrates and products. The flow rate was 1 mL/
min. The column temperature was ambient, and the detection
wavelength was 220 nm. The retention times for 2 and 4 were
activity were pooled. A unit of activity was defined as mmol of
product formed per min per mg protein.
1
4.8 min and 6.7 min, respectively.
Resolution of enantiomers of Cbz-amino acids was carried
Sodium Dodecyl Sulfate Polyacrylamide Gel
Electrophoresis (SDS/PAGE)
out by using a Chiralcel OJ-RH column (5 mm; 4.6 Â 150 mm).
The column was equilibrated with 0.05% TFA in methanol:
water (20:80). A gradient in 30 min with solvent A [0.05%
TFA in methanol:water (20:80)ꢀ running from 100 ± 0% and
solvent B [0.05% TFA in methanol: acetonitrile (20:80)ꢀ
running from 0 ± 100% was applied at 0.8 mL/min flow rate.
The detector was set up at 220 nm. Retention times were: Cbz-
d-phenylalanine, 21.06 min; Cbz-l-phenylalanine, 21.69 min;
Cbz-d-tyrosine, 19.15 min; Cbz-l-tyrosine 19.68 min; Cbz-d-
proline, 14.85 min, Cbz-l-proline 15.41 min.
¾
The active fractions from the Sephacryl S-200 column were
¾
evaluated by SDS-PAGE as described in the PhastSystem
procedure by Pharmacia[ using the homogeneous 12.5%
Phastgel. The enzyme samples (10 mL containing 10 mg
protein) were added to a buffer containing 10 mM Tris-HCl,
1 mM EDTA, pH 8, 2.5% SDS and 5% b-mercaptoethanol.
The mixture was heated at 1008C for 5 minutes, and
bromophenol blue was added to 0.01%. Gels were stained
with silver stain and destained in 10% acetic acid solution.
Markers with standard molecular weights were phosphorylase
b (94,000), bovine serum albumin (67,000), ovalbumin
21ꢀ
Resolution of enantiomers of amino acids was carried out by
using a Regis Ligand Exchange Column (5 mm; 4.6 Â 150 mm)
Adv. Synth. Catal. 2003, 345, 830 ± 834
asc.wiley-vch.de
¹ 2003 WILEY-VCH Verlag GmbH & Co. KGaA, Weinheim
833

 Patel, Ramesh N.
Patel, Ramesh N.
 Nanduri, Venkata
Nanduri, Venkata
 Brzozowski, David
Brzozowski, David
 McNamee, Clyde
McNamee, Clyde
 Banerjee, Amit
Banerjee, Amit