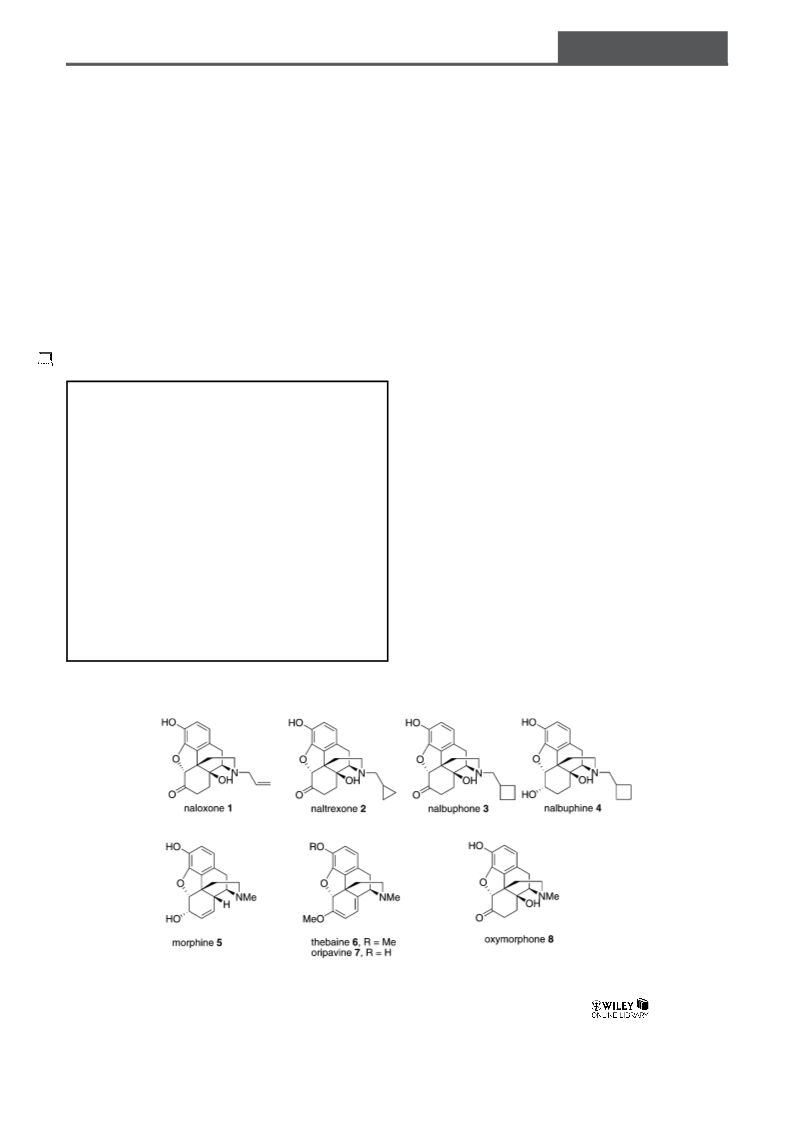
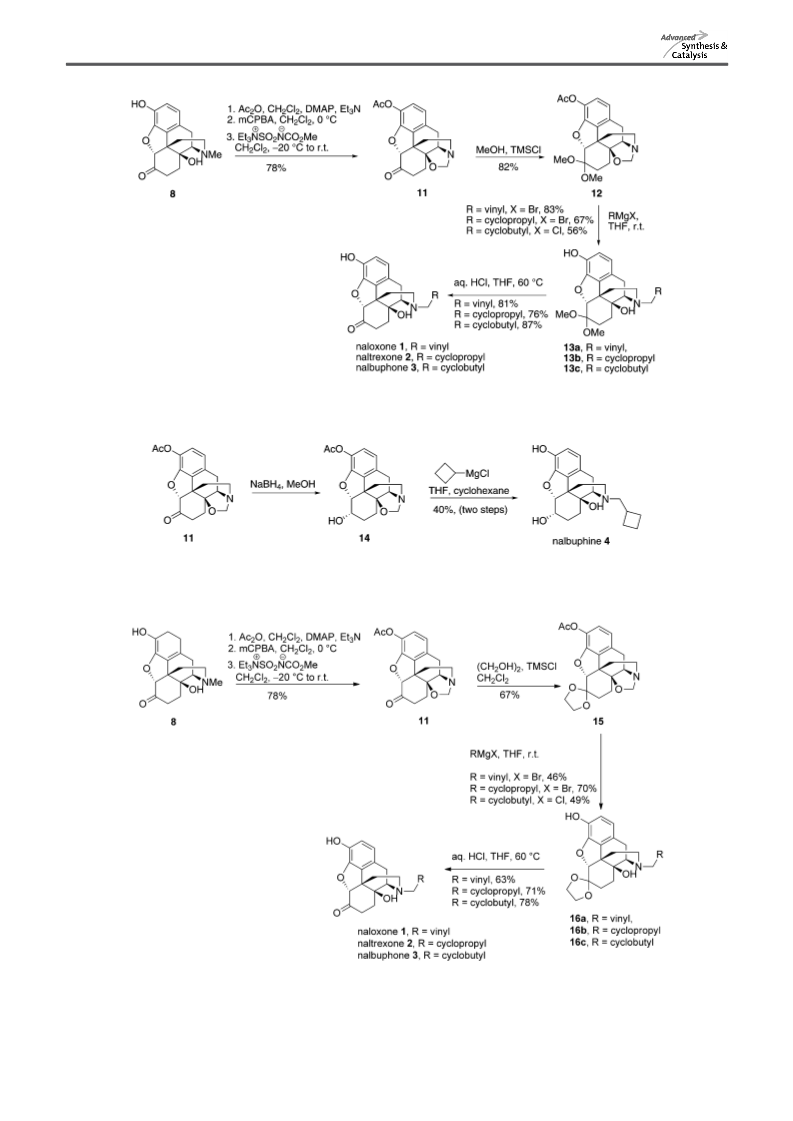
General Method of Synthesis for Naloxone, Naltrexone, Nalbuphone, and Nalbuphine
À
À
À
À
(O CH), 70.4 (HO C), 62.3 (N CH), 57.7 (N CH2CH),
H. Leisch, A. Moudra, B. Allen, V. De Luca, D. P. Cox,
T. Hudlicky, Collect. Czech. Chem. Commun. 2009, 74,
1179; d) for a review on O- and N-demethylase en-
zymes, see: J. M. Hagel, P. J. Facchini, Front. Plant
Physiol. 2010, 1, 14.
À
À
51.0 (C), 43.3 (N CH2CH2), 36.2 (O=C CH2), 31.3
À
À
(HO C- CH2), 30.5 (NCH2CH2), 22.7 (PhCH2).
[9] N-Demethylation of N-oxides with iron, see: a) G.
Kok, T. D. Asten, P. J. Scammells, Adv. Synth. Catal.
2009, 351, 283; b) Z. Dong, P. J. Scammells, J. Org.
Chem. 2007, 72, 9881; c) G. B. Kok, C. C. Pye, R. D.
Singer, P. J. Scammells, J. Org. Chem. 2010, 75, 4806;
d) G. B. Kok, P. J. Scammells, Bioorg. Med. Chem. Lett.
2010, 20, 4499; e) C. Smith, S. Purcell, L. Waddell, N.
Hayes, J. Ritchie, Patent WO 2005/028483, 2005.
[10] N-Demethylation of quaternary salts, see: T. Hudlicky,
T. R. Carroll, H. Leisch, A. Machara, L. Werner, D. R.
Adams, PCT Int. Appl. PCT/CA2010/000587, 2010;
CODEN: PIXXD2 WO 2010121369A1 20101028 CAN
153:555396 AN 2010:1342585.
Acknowledgements
The authors are grateful to the following agencies for finan-
cial support of this work: Natural Sciences and Engineering
Research Council of Canada (NSERC) (Idea to Innovation
and Discovery Grants); Canada Research Chair Program,
Canada Foundation for Innovation (CFI), Research Corpo-
ration, TDC Research, Inc., TDC Research Foundation, and
Brock University.
References
[11] N-Demethylation of quaternary salts (nalbuphine syn-
thesis), see: A. Machara, D. P. Cox, T. Hudlicky, Het-
erocycles 2012, 84, 615.
[12] N-Demethylation of quaternary salts (buprenorphine
synthesis), see: L. Werner, A. Machara, D. R. Adams,
D. P. Cox, T. Hudlicky, J. Org. Chem. 2011, 76, 4628.
[13] Pd-catalyzed N-demethylation/acylation, see: T. Hud-
licky, A. Machara, US Provisional Patent Application
No: 61/483,264. Filing Date: May 6, 2011.
[14] Pd-catalyzed N-demethylation/acylation (naltrexone
synthesis), see: A. Machara, D. P. Cox, T. Hudlicky,
Adv. Synth. Catal. 2012, 354, 2713.
[15] T. Hudlicky, A. Machara, L. Werner, M. Wernerova,
M. A. Endoma-Arias, US Provisional Patent Applica-
tion No: 61/489,359. Filing Date: May 2, 2011.
[1] For recent reviews on the synthesis and/or pharmacolo-
gy of opiate-derived pharmaceuticals see: a) P. Lobmai-
er, M. Gossop, H. Waal, J. Bramness, Eur. J. Clin. Phar-
macol. 2010, 66, 537; b) H. Schmidhammer, M. Spetea,
Top. Curr. Chem. 2011, 299, 63; c) I. Kissin, Anesthesi-
ology 2010, 110, 780.
[2] For detailed list of references to the activities and me-
dicinal use of these compounds see: L. Werner, M.
Wernerova, A. Machara, M. A. Endoma-Arias, J.
Duchek, D. R. Adams, D. P. Cox, T. Hudlicky, Adv.
Synth. Catal. 2012, 354, 2706.
[3] a) P. X. Wang, J. Tao, D. W. Berberich, U.S. Patent
8,080,661, 2009; b) O. H. Kvernenes, A. M. Nygard, A.
Heggelund, H. Halvorson, Patent WO2007137785A2,
2007.
[16] A. Machara, T. Hudlicky, unpublished observations.
[17] For examples of nucleophilic opening of oxazolidines
see: with cuprates: a) T. Kobayashi, F. Hasegawa, Y.
Hirose, K. Tanaka, S. Katsumura, H. Mori, J. Org.
Chem. 2012, 77, 1812; b) P. Viaud, V. Coeffard, C.
Thobie-Gautier, I. Beaudet, N. Balland, J.-P. Quintard,
E. Le Grognec, Org. Lett. 2012, 14, 942; c) T. Kobaya-
shi, F. Hasegawa, K. Tanaka, S. Kasumura, Org. Lett.
2006, 8, 3813; with Grignard reagents: d) G. Arena, J.
Salvadori, M. Taddei, N. Zill, N. Girard, A. Mann, Org.
Lett. 2011, 13, 2294; e) J. Alladoum, S. Roland, E.
Vrancken, P. Mangeney, C. Kadouri-Puchot, J. Org.
Chem. 2008, 73, 9771; with Grignard reagents and
BF3·Et2O: f) R. Noel, C. Vanucci-Bacque, M.-C. Far-
geau-Bellassoued, G. Lhommet, Eur. J. Org. Chem.
2007, 3, 476; g) by organolithiums: L. J. Simon, E. Che-
lain, T. Brigaud, Org. Lett. 2012, 14, 604.
[4] a) N. J. Goodwin, M. Mitchell, N. K. Thomson, G. W.
Wilson, M. J. Young, Patent WO2006035195A1, 2006;
b) E. C.
Hudson,
S.
Woods,
U.S.
Patent
20100210843A1, 2010.
[5] N-Demethylation with CNBr, see: J. von Braun, Ber.
Dtsch. Chem. Ges. 1900, 33, 1438.
[6] N-Demethylation with chloroformates, see: a) J. H.
Cooley, E. J. Evain, Synthesis 1989, 1; b) R. A. Olofson,
J. T. Martz, J. P. Senet, M. Piteau, T. J. Malfroot, J. Org.
Chem. 1984, 49, 2081.
[7] N-Demethylation with l-Selectride, see: a) A. Coop,
J. W. Janetka, J. W. Lewis, K. C. Rice, J. Org. Chem.
1998, 63, 4392–4396; see also: b) G. Majetich, Y.
Zhang, K. Wheless, Tetrahedron Lett. 1994, 35, 8727–
8730.
[8] N-Demethylation by biological/enzymatic methods,
see: a) K. M. Madyastha, Proc. Indian Acad. Sci. 1994,
106, 1203; b) K. M. Madyastha, G. V. B. Reddy, J.
Chem. Soc. Perkin Trans. 1 1994, 911; c) V. Chaudhary,
[18] J.-D. Andre, J.-R. Dormoy, A. Haymes, Synth.
Commun. 1992, 22, 2313.
Adv. Synth. Catal. 0000, 000, 0 – 0
ꢀ 2013 Wiley-VCH Verlag GmbH & Co. KGaA, Weinheim
5
ÞÞ
These are not the final page numbers!

 Endoma-Arias, Mary Ann A.
Endoma-Arias, Mary Ann A.
 Cox, D. Phillip
Cox, D. Phillip
 Hudlicky, Tomas
Hudlicky, Tomas