Angewandte Chemie International Edition
10.1002/anie.201712852
COMMUNICATION
[4]
5]
A. J. Dixon, M. J. McGrath, P. O’Brien, Org. Synth. 2006, 83, 141-154.
a) P. Beak, A. Basu, D. J. Gallagher, Y. S. Park, S. Thayumanavan, Acc.
Chem. Res. 1996, 29, 552-560; b) D. Hoppe, T. Hense, Angew. Chem.
Int. Ed. Engl. 1997, 36, 2282-2316; Angew. Chem. 1997, 109, 2376-
Tollari, Synthesis 1984, 4, 353-356; c) K. Paulvannan, J. R. Stille, J. Org.
Chem. 1992, 57, 5319-5328.
[
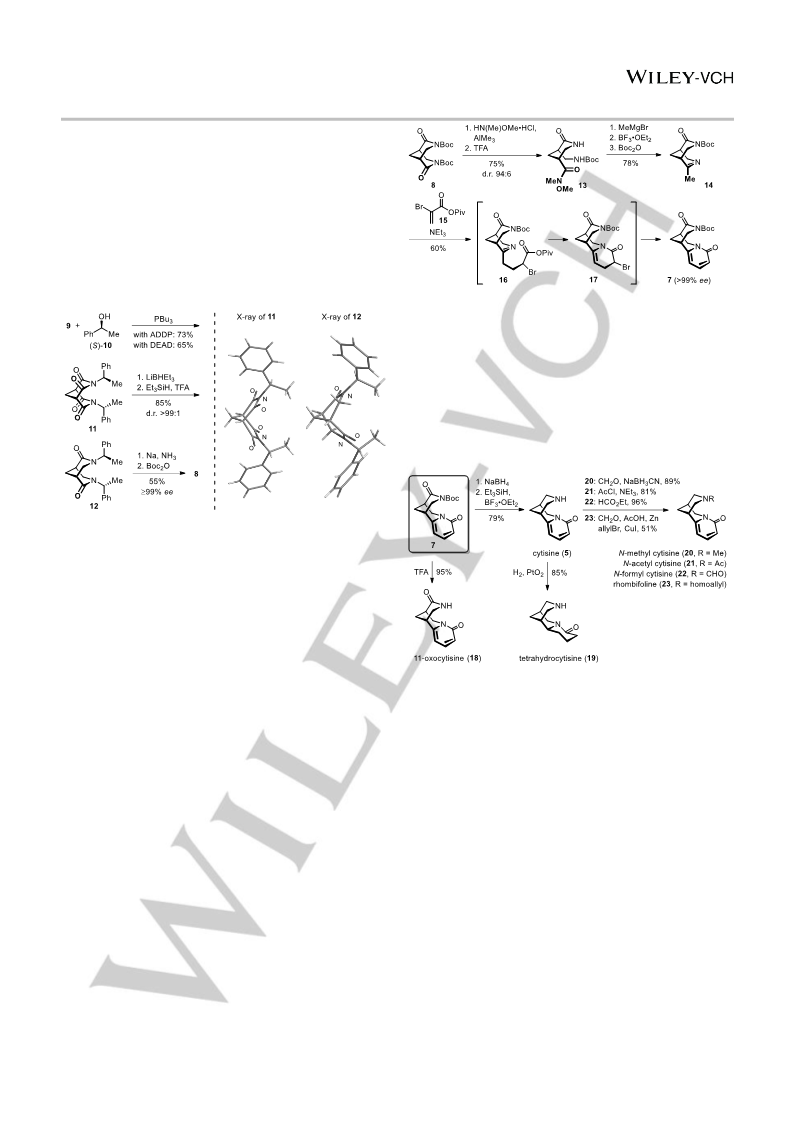
[16] Crude 7 had just 94% ee. The loss of enantiopurity is most likely caused
by the minor trans-diastereomer of 13, which could not be removed. It
had to be carried through the following steps, providing a mixture of 7
and ent-7. The enantiopurity of 7 was re-raised to >99% by simple
fractional crystallization.
2
410; c) D. Hoppe, G. Christoph in The Chemistry of Organolithium
Compounds, Vol. 2 (Eds.: Z. Rappoport, I. Marek), Wiley, Chichester,
004, pp. 1055-1164; d) O. Chuzel, O. Riant in Topics in Organometallic
2
Chemistry, Vol 15: Chiral Diazaligands for Asymmetric Synthesis (Eds.:
M. Lemaire, P. Mangeney), Springer, Berlin, 2005, pp. 59-92; e) J.-C.
Kizirian, Top. Stereochem. 2010, 26, 189-251; f) E. A. Mitchell, A.
Peschiulli, N. Lefevre, L. Meerpoel, B. U. W. Maes, Chem. Eur. J. 2012,
[17] a) Tetrahydrocytisine (19): M. J. Johansson, L. Schwartz, M. Amedjkouh,
N. Kann, Tetrahedron: Asymmetry 2004, 15, 3531-3538; b) N-methyl
cytisine (20): F. Frigerio, C. A. Haseler, T. Gallagher, Synlett 2010, 5,
729-730; c) N-acetyl cytisine (21): J. Rouden, A. Ragot, S. Gouault, D.
Cahard, J.-C. Plaquevent, M.-C. Lasne, Tetrahedron: Asymmetry 2002,
13, 1299-1305; d) N-formyl cytisine (22): S. Ohmiya, H. Otomasu, I.
Murakoshi, J. Haginiwa, Phytochemistry 1974, 13, 643-644.
[18] M. Breuning, D. Hein, M. Steiner, V. H. Gessner, C. Strohmann, Chem.
Eur. J. 2009, 15, 12764-12769.
1
8, 10092-10142.
S. P. Thomas, R. M. French, V. Jheengut, V. K. Aggarwal, Chem. Rec.
009, 9, 24-39.
[
6]
7]
2
[
a) M. S. Sigman, D. R. Jensen, Acc. Chem. Res. 2006, 39, 221-229; b)
D. C. Ebner, R. M. Trend, C. Genet, M. J. McGrath, P. O’Brien, B. M.
Stoltz, Angew. Chem. Int. Ed. 2008, 47, 6367-6370; Angew. Chem. 2008,
[19] M. Breuning, M. Steiner, C. Mehler, A. Paasche, D. Hein, J. Org. Chem.
2009, 74, 1407-1410.
120, 6467-6470.
[
8]
9]
For bispidine derivatives in enantioselective, copper-catalyzed Henry
reactions, see: a) H. Maheswaran, K. L. Prasanth, G. G. Krishna, K.
Ravikumar, B. Sridhar, M. L. Kantam, Chem. Commun. 2006, 4066-
[20] a) H. Kubo, S. Ohmiya, I. Murakoshi, Can. J. Chem. 1994, 72, 214-217;
b) J. R. Harrison, P. O’Brien, Tetrahedron Lett. 2000, 41, 6167-6170; c)
A. Consonni, B. Danieli, G. Lesma, D. Passarella, P. Piacenti, A. Silvani,
Eur. J. Org. Chem. 2001, 1377-1383; d) J. Kurek, B. Jasiewicz, E.
Wyrzykiewicz, W. Boczoń, J. Mol. Struct. 2011, 1003, 10-20; e) P. R.
Blakemore, N. R. Norcross, S. L. Warriner, P. C. Astles, Heterocycles
2006, 70, 609-617.
[21] The 1H and 13C NMR spectra of 25 and 32 display two sets of signals,
those of 36 three sets of signals. They probably result from N-Boc
rotamers of the exo-configured diastereomer, but the existence of endo-
diastereomers cannot be fully excluded.
4068; b) J. Canipa, A. Stute, P. O’Brien, Tetrahedron 2014, 70, 7395-
7403; c) D. Scharnagel, A. Müller, F. Prause, M. Eck, J. Goller, W. Milius,
M. Breuning, Chem. Eur. J. 2015, 21, 12488-12500.
[
For the enantioselective total synthesis of bispidine natural products,
see: a) (+)-sparteine (ent-1): B. T. Smith, J. A. Wendt, J. Aubé, Org. Lett.
2
002, 4, 2577-2579; b) (–)-sparteine (1): J.-P. R. Hermet, M. J. McGrath,
P. O’Brien, D. W. Porter, J. Gilday, Chem. Commun. 2004, 16, 1830-
831; c) J. D. Firth, S. J. Canipa, L. Ferris, P. O'Brien, Angew. Chem. Int.
1
Ed. 2018, 57, 223–226; Angew. Chem. 2018, 130, 229–232; d) (–)-
cytisine (5): B. Danieli, G. Lesma, D. Passarella, A. Sacchetti, A. Silvani,
A. Virdis, Org. Lett. 2004, 6, 493-496; e) (+)-cytisine (ent-5): T. Honda,
R. Takahashi, H. Namiki, J. Org. Chem. 2005, 70, 499-504; f) D. Gray,
T. Gallagher, Angew. Chem. Int. Ed. 2006, 45, 2419-2423; Angew.
Chem. 2006, 118, 2479–2483; g) (+)- and (–)-cytisine (5 and ent-5): F.
R. Struth, C. Hirschhäuser, Eur. J. Org. Chem. 2016, 958-964; h) (+)-
thermopsine (ent-24): R. Brimioulle, T. Bach, Science 2013, 342, 840-
[22] Note that tetrahydrorhombifoline (30) and rhombifoline (23) possess
'enantiomeric' bispidine cores.
2
[23] Twofold reduction of 8 with Cp ZrHCl was found to give higher yields, as
compared to NaBH , LiBHEt , or iBu
4
3
2
AlH.
[24] The Lewis acid assisted addition of 4-chlorobutylzinc bromide worked
well on the bis-N,O-acetal 36 (Scheme 7), but failed for unknown reasons
on 25 (Scheme 5) and 32 (Scheme 6).
843; i) (+)--isosparteine (ent-37): F. M. Al-Saffar, R. C. D. Brown, Org.
Lett. 2017, 19, 3502-3504; j) virgiboidine: S. Kress, J. Weckesser, S. R.
Schulz, S. Blechert, Eur. J. Org. Chem. 2013, 1346-1355.
[
10] An earlier attempt to desymmetrize the N,N'-dibenzyl or N,N'-diallyl
derivative of 9 by CBS reduction had little success, see: N. R. Norcross,
J. P. Melbardis, M. F. Solera, M. A. Sephton, C. Kilner, L. N. Zakharov,
P. C. Astles, S. L. Warriner, P. R. Blakemore, J. Org. Chem. 2008, 73,
7
939-7951.
11] P. R. Blakemore, C. Kilner, N. R. Norcross, P. C. Astles, Org. Lett. 2005,
, 4721-4724.
[
[
7
12] The sense and level of asymmetric induction is surprising and cannot be
explained by simple steric effects. All preferred conformations, in which
the phenyl group of the chiral auxiliary will occupy an exo position (cp.
crystal structure of 11), suggest a weak shielding of the attacked pro-S
carbonyl groups, but not of the untouched pro-R carbonyl groups. For a
related reduction of an N-phenylethyl substituted, norbornane fused
succinimide giving the same unexpected stereochemical outcome, see:
P. Rackler, C. Berthold, F. Voss, T. Bach, J. Am. Chem. Soc. 2010, 132,
1
5911.
13] Crystals of 11 and 12 were obtained from Et
815735 (11) and 1565771 (12) contain the supplementary
[
[
[
2 2 2
O/CH Cl /pentane. CCDC
1
crystallographic data for this paper. These data can be obtained free of
14] All attempts to selectively modify just one carbonyl group in 8, while
keeping the bicyclic system intact (e.g., N-Boc deprotection or carbonyl
reduction), mainly provided the undesired product of an attack on both
imides. Ring opening reactions, by contrast, occurred with high mono-
selectivity, presumably due to the release of conformational strain.
15] For the construction of dihydropyridones from imines and acrylic acid
derivatives see, for example: a) P. W. Hickmott, G. Sheppard, J. Chem.
Soc. C 1971, 1358-1362; b) B. Danieli, G. Lesma, G. Palmisano, S.
This article is protected by copyright. All rights reserved.

 Scharnagel, Dagmar
Scharnagel, Dagmar
 Goller, Jessica
Goller, Jessica
 Deibl, Nicklas
Deibl, Nicklas
 Milius, Wolfgang
Milius, Wolfgang
 Breuning, Matthias
Breuning, Matthias