New Route to Silicon Alkoxides from Silica
1035
2
3
to silicon). B.p. 168 C. 29Si n.m.r. (CDCl3) 82. Accurate
mass: 193 089481 [C7H17O4Si (M CH3) requires 193 089613].
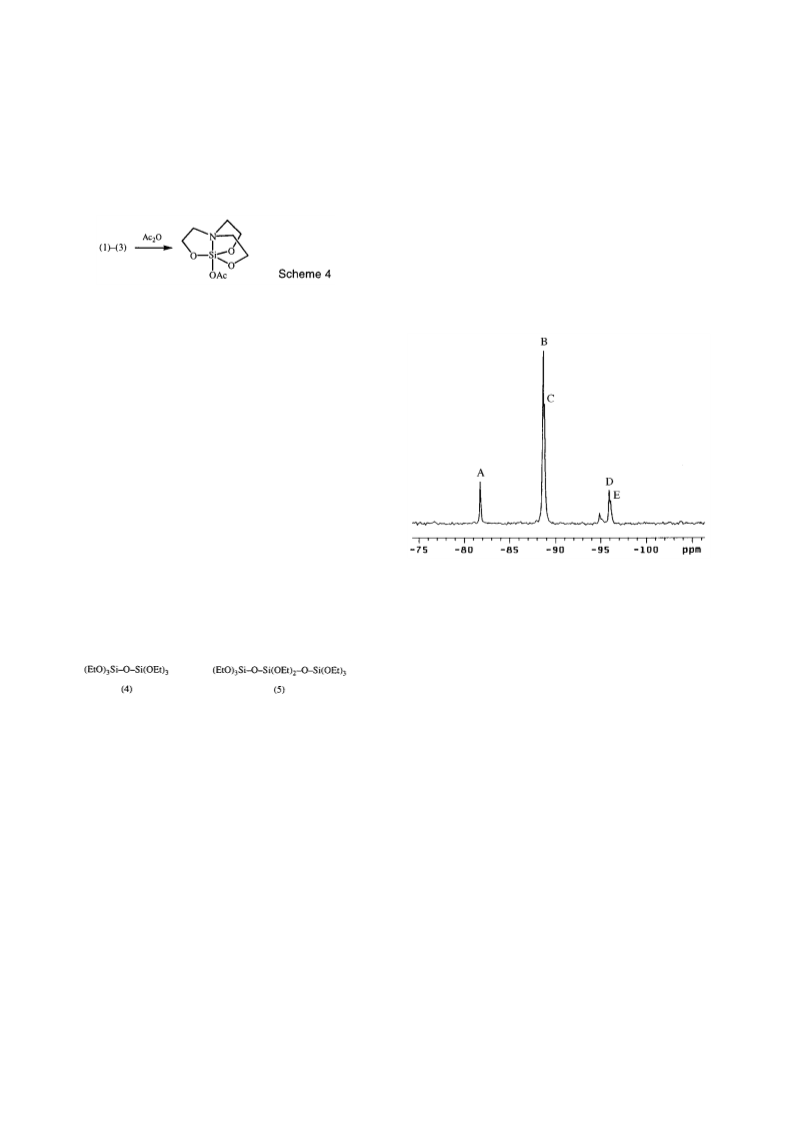
Compound (4) (8 9 g, 72%). B.p. 77 C/1 5 mmHg. 29Si n.m.r.
(CDCl3) 88 87. Accurate mass: 342 153633 [C12H30O7S2
(M)+ requires 342 153010]. Compound (5) (0 5 g, 4%). B.p.
92 C/1 0 mmHg. 29Si n.m.r. (CDCl3) 88 9, 95 9. Accu-
rate mass: 431 159075 [C14H35O9S13 (M OC2H5)+ requires
431 158893]. A small amount of involatile residue, presumed
to be higher polysiloxanes, remained in the distillation ask.
Goodwin, G. B., and Kenney, M. E., in ‘Silicon-Based
Polymer Science’ (Eds J. M. Zeigler and F. W. Gordon
Fearon) Vol. 15, pp. 251–263 (American Chemical Society:
Washington, DC, 1990).
Goodwin, G. B., and Kenney, M. E., in ‘Inorganic and
Organometallic Polymers’ (Eds M. Zeldin, K. J. Wynne and
H. R. Allcock) Vol. 18, pp. 238–248 (American Chemical
Society: Washington, DC, 1988).
Suzuki, E., Akiyama, M., and Ono, Y., J. Chem. Soc.,
Chem. Commun., 1992, 136.
Rosenheim, A., Raibmann, B., and Schendel, G., Z. Anorg.
Chem., 1931, 196, 160.
Barnum, D. W., Inorg. Chem., 1972, 11, 1424.
Laine, R. M., Blohowiak, K. Y., Robinson, T. R., Hoppe,
M. L., Nardi, P., Kampf, J., and Uhm, J., Nature (London),
1991, 353, 642.
Blohowiak, K. Y., Treadwell, D. R., Mueller, B. L., Hoppe,
M. L., Jouppi, S., Kansal, P., Chew, K. W., Scotto, C. L.
S., Babonneau, F., and Kampf, J., Chem. Mater., 1994, 6,
2177.
Gainsford, G. J., Kemmitt, T., and Milestone, N. B., Acta
Crystallogr., Sect. C, 1995, 51, 8.
Boudin, A., Cerveau, G., Chuit, C., Corriu, R. J. P., and
Reye, C., Angew. Chem., Int. Ed. Engl., 1986, 25, 474.
Kemmitt, T., unpublished data, 1995.
Zhang, Z., Hoppe, M. L., Rahn, J. A., Koo, S., and Laine,
R. M., Mater. Res. Soc. Symp. Proc., 1992, 249, 107.
Kansal, P., and Laine, R. M., J. Am. Ceram. Soc., 1994,
77, 875.
Kansal, P., and Laine, R. M., J. Am. Ceram. Soc., 1995,
78, 529.
Kemmitt, T., and Milestone, N. B., in ‘Silicon Containing
Polymers’ (Ed. R. G. Jones) pp. 107–112 (Royal Society
of Chemistry: Cambridge 1995).
Laine, R. M., Treadwell, D. R., Mueller, B. L., Bickmore,
C., Waldner, K. F., and Hinklin, T. R., J. Mater. Chem.,
1996, 6, 1441.
Waldner, K. F., Laine, R. M., Dhumrongvaraporn, S.,
Tayaniphan, S., and Narayanan, R., Chem. Mater., 1996,
8, 2850.
Frye, C. L., Vincent, G. A., and Finzel, W. A., J. Am.
Chem. Soc., 1971, 93, 6805.
Kemmitt, T., and Milestone, N. B., Industrial Research
Ltd Report 345, Wellington, New Zealand, 1995.
Kemmitt, T., and Henderson, W, J. Chem. Soc., Perkin
Trans. 1, 1997, 729.
Frye, C. L., Vogel, G. E., and Hall, J. A., J. Am. Chem.
Soc., 1961, 83, 996.
4
5
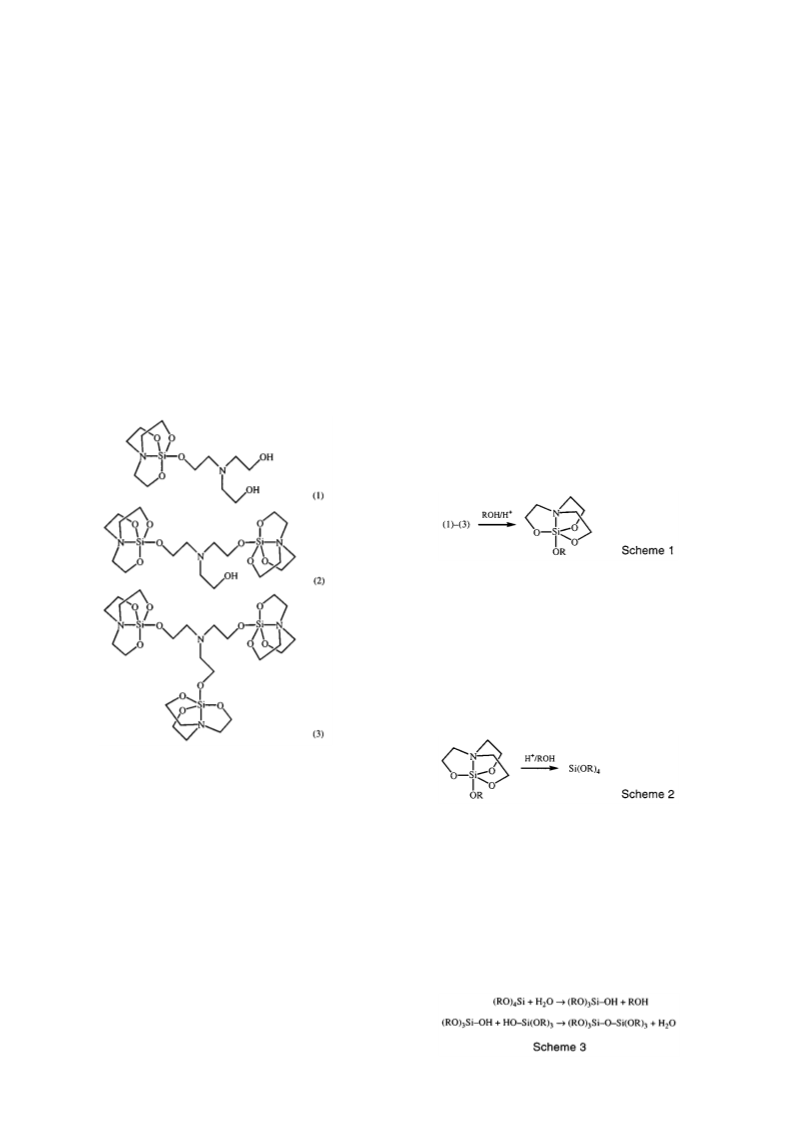
(g) Tetramethoxysilane
In a similar method to (f) above, methoxysiloxanes were
prepared from triethanolamine-substituted silatranes as formed
in (b) above, in acidic methanol (100 ml) solution. Si(OMe)4
was isolated in 38% yield. B.p. 121 C. 1H n.m.r. (CDCl3) 3 6.
13C n.m.r. (CDCl3) 51 1. 29Si n.m.r. (CDCl3) 78 2. The
other major product was identi ed as hexamethoxydisiloxane
(33%). 29Si n.m.r. (CDCl3) 78 2.
6
7
8
9
(h) Acetoxysilatrane
Excess acetic anhydride (20 ml) was added to a sample
of triethanolamine-substituted silatranes (38 8 g, 0 139 mol)
[formulated from elemental analysis as Si(tea)1 72] as formed
in (b) above. The ask was warmed gently to melt the
silatrane and assist in the dissolution. The reaction mixture
was re uxed for 0 5 h before cooling to room temperature
to lter o the o -white product. Recrystallization from hot
propan-2-ol produced a white crystalline product [23 9 g, 74%
yield ( rst crop)] (Found: C, 41 1; H, 6 3; N, 5 9. C8H15NO5Si
10
11
12
13
14
15
requires C, 41 2; H, 6 5; N, 6 0%). 1H n.m.r.
(CDCl3)
1 80, s, CH3CO2; 2 88, t, CH2N; 3 68, t, CH2O. 13C n.m.r.
(CDCl3) 23 41, CH3CO; 52 12, CH2N; 58 54, CH2O; 174 01,
CH3CO. 29Si n.m.r. (CDCl3) 99 9.
16
17
Conclusion
The reactivity of amorphous hydrated silica makes
it a convenient starting material for reaction with
triethanolamine, particularly when used in conjunction
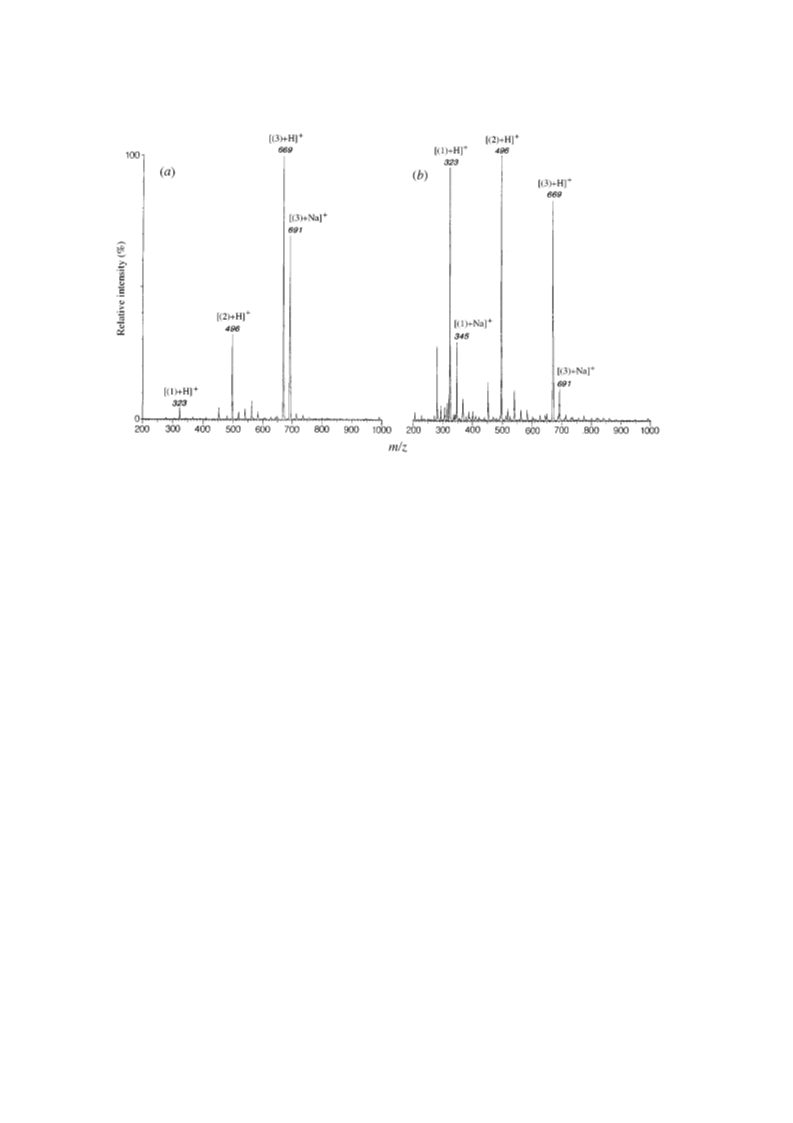
with an alkali metal base catalyst. Electrospray mass
spectrometry proved to be extremely useful in char-
acterizing the silatranyl products. Further reaction of
the silatranes in acidic alcohols resulted in alkoxide
exchange to yield alkoxysilanes. The method described
represents an alternative route to Si(OEt)4 and other
alkoxysilanes from a readily available silica resource.
18
19
20
21
22
23
Voronkov, M. G., Dyakov, V. M., and Kirpichenko, S. V.,
J. Organomet. Chem., 1982, 233, 1.
Garant, R. J., Daniels, L. M., Das, S. K., Janakiraman,
M. N., Jacobson, R. A., and Verkade, J. G., J. Am. Chem.
Soc., 1991, 113, 5728.
Bonchio, M., Licini, G., Modena, G., Moro, S., Bortolini,
O., Traldi, P., and Nugent, W. A., Chem. Commun., 1997,
869.
References
24
1
Kirk–Othmer Encyclopedia of Chemical Technology, 3rd
Edn, Vol. 20, p. 750 (Wiley–Interscience: New York 1979).

 Kemmitt, Tim
Kemmitt, Tim
 Henderson, William
Henderson, William