1
514
Vol. 53, No. 11
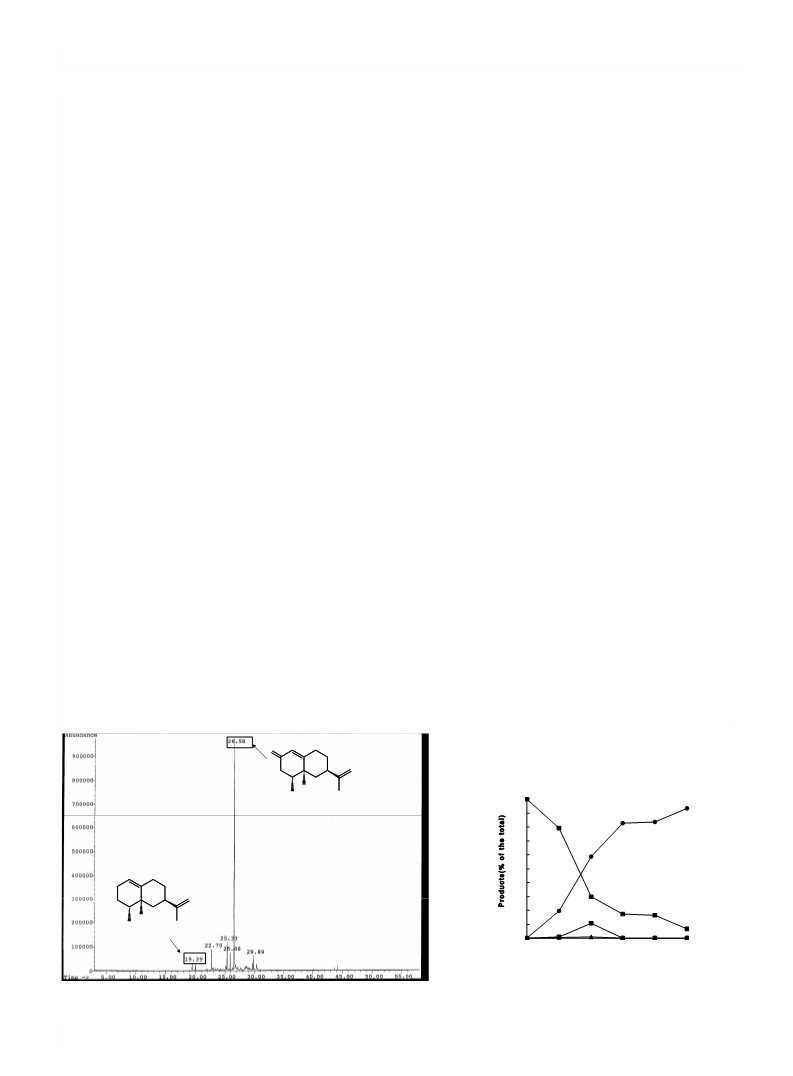
Table 1. Conversion of Valencene (1) to Nootkatone (2) by Chlorella Table 3. Biotransformation of (ꢀ)-Valecene (1) by Botryodiplodia theo-
Species for 14 d
bromae (in GC-MS)
Metabolites (% of the total in GC-MS)
Metabolites (% of the total in GC-MS)
Conversion
ratio (%)
Botryodiplodia
theobromae
Conversion
ratio (%)
Chlorella sp.
1
3
4
2
1
3
4
2
C. fusuca
C. pyrenoidosa
C. vulgaris
11
7
0
0
0
0
0
0
0
89
93
100
89
93
100
Sudachi BT8603
Pear BT8002
28
56
11
0
20
9
42
35
72
44
Compounds: 1: (ꢀ)-valencene; 2: (ꢀ)-nootkatone; 3: 2a-hydroxyvalencene; 4:
nootkatol.
medium and incubated for a further 7 d to obtain the metabo-
lites. Only the results from five strains characteristic of the
31 strains are shown in Tables 2 and 3.
In summary, the expensive grapefruit aromatic, nootkatone
(2) used by cosmetic and fiber manufacturers was obtained in
excellent yield by biotransformation of (ꢀ)-valencene (1),
which can be cheaply obtained from Valencia oranges, by
Chlorella species, fungi such as Mucor species, B. dothidea,
and B. theobromae. This is a very inexpensive and clean oxi-
dation reaction that dose not use any heavy metals, and thus
this method is expected to find applications in the industrial
production of nootkatone.
Acknowledgments We thank Dr. M. Tanaka (TBU) and Miss Y.
Okamoto (TBU) for providing 600-MHz NMR and mass spectra and Ms. C.
Murakami for technical assistance. We thank Takasago International Co.,
Ltd., Japan, for providing valencene and nootkatone.
References
1)
Kieslich K., Annual Reports on Fermentation Processes, 1, 267—297
1977).
Fig. 3. Possible Metabolic Pathways of Valencene (1) by Chlorella species
(
2
)
Noma Y., Asakawa Y., “Biotechnology in Agriculture and Forestry,”
Vol. 28, ed. by Bajaj Y. P. S., Springer, Berlin, 1994, p. 185.
Table 2. Biotransformation of (ꢀ)-Valencene (1) by Botryosphaeria doth-
idea (in GC-MS)
3) Noma Y., Asakawa Y., “Biotechnology in Agriculture and Forestry,”
Vol. 33, ed. by Bajaj Y. P. S., Springer, Berlin, 1995, p. 62.
4) Noma Y., Asakawa Y., “Biotechnology in Agriculture and Forestry,”
Vol. 41, ed. by Bajaj Y. P. S., Springer, Berlin, 1998, p. 194.
5) Hashimoto T., Noma Y., Kato S., Tanaka M., Takaoka S., Asakawa Y.,
Chem. Pharm. Bull., 47, 716—717 (1999).
Metabolites (% of the total in GC-MS)
Botryosphaeria
dothidea
Conversion
ratio (%)
1
3
4
2
Peach PP8402
Ume BD8398II
Rose BD8303II
0
8
3
15
18
8
21
31
5
64
43
84
100
92
97
6) Lahlou E. H., Noma Y., Hashimoto T., Asakawa Y., Phytochemistry,
54, 455—460 (2000).
7) Hashimoto T., Noma Y., Gotoh Y., Tanaka M., Takaoka S., Asakawa
Y., Heterocycles, 62, 655—666 (2004).
Compounds: 1: (ꢀ)-valencene; 2: (ꢀ)-nootkatone; 3: 2a-hydroxyvalencene; 4:
nootkatol.
8)
Hashimoto T., Noma Y., Asakawa Y., Heterocycles, 54, 529—559
2001).
Matsumoto T., Hayashi N., Ishida T., Asakawa Y., J. Pharm. Sci., 79,
40—547 (1990).
0) Matsumoto T., Hayashi N., Ishida T., Asakawa Y., Chem. Pharm. Bull.,
0, 1721—1726 (1992).
1) Haze S., Sakai K., Gozu Y., Jpn. J. Pharmacol., 90, 247—253 (2002).
(
9)
5
A fungus strain from the soil adhering to the liverwort
Pallavicinia subcilita was identified as Mucor sp., which was
inoculated and cultivated while stationary in Czapek-pepton
1
1
4
medium (pH 7.0) at 30 °C for 7 d. Compound 1 (20 mg/ 12) Dhavalikar R. S., Albroscheit G., Dragoco Rep., 20, 251—258 (1979).
1
1
3) Okuda M., Sonohara K., Takikawa H., Jpn. Kokai Tokkyo Koho,
03967 (1994).
4) Sowden R. J., Yasmin S., Rees N. H., Bell S. G., Luet-Lok Wong.,
5
7
0 ml) was added to the medium and incubated for a further
d. Nootkatone (2) was then obtained in very high yield
3
(82%).
Org. Biomol. Chem., 3, 57—64 (2005).
Next, the biotransformation from 1 to 2 was examined 15) Wilson C. W., III, Shaw P. E., J. Agric. Food Chem., 26, 1430—1432
using the plant pathogenic fungi Botryosphaeria dothidea
and Botryodiplodia theobromae (a total of 31 strains) sepa-
rated from fungi infecting various types of fruit, etc. B. doth-
idea and B. theobromae were both inoculated and cultivated
while stationary in Czapek-pepton medium (pH 7.0) at 30 °C
for 7 d. The substrate 1 (20 mg/50 ml) was added to each
(1978).
1
1
6) Salvador J. A. R., Clark J. H., Green Chemistry, 4, 352—356 (2002).
7) Hashimoto T., Asakawa Y., Noma Y., Murakami C., Tanaka M., Kani-
sawa T., Emura M., Jpn. Kokai Tokkyo Koho, 70492A (2003).
8) Hashimoto T., Asakawa Y., Noma Y., Murakami C., Furusawa M.,
Kanisawa T., Emura M., Mitsuhashi K., Jpn. Kokai Tokkyo Koho,
250591A (2003).
1

 Furusawa, Mai
Furusawa, Mai
 Hashimoto, Toshihiro
Hashimoto, Toshihiro
 Noma, Yoshiaki
Noma, Yoshiaki
 Asakawa, Yoshinori
Asakawa, Yoshinori