2
. . . . . . . . . . . . . . . . . . . . . . . . . . . . Zada et al. Sci China Chem . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 2
rice, potato and corn. [9]. However, it would be more ap-
propriate to use non-edible precursors such as cellulose and
di-saccharides for the synthesis of sorbitol [9,10]. In this
regard, the non-edible cellobiose, which could produce with
high yield from cellulose [11,12], becomes an ideal substrate
for the synthesis of sorbitol.
with the mono-metallic catalysts, the superior performance
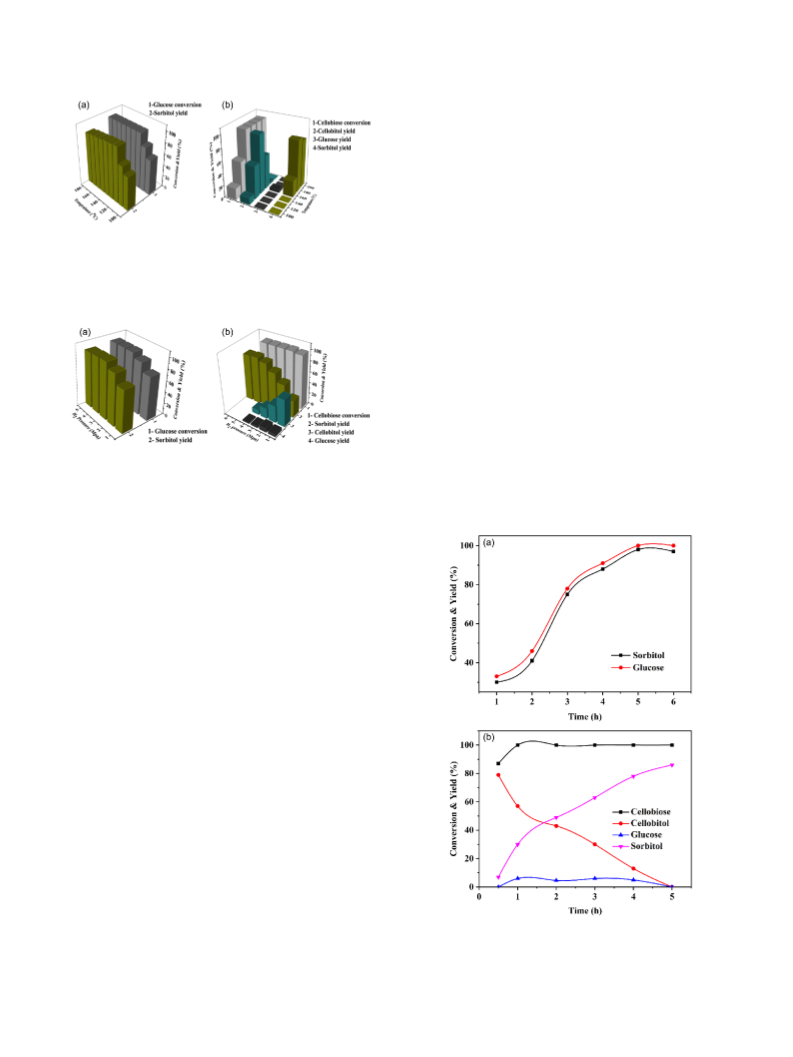
was possibly due to the alloy species. In the kinetic studies,
cellobitol was detected as the key intermediate. Furthermore,
NiCo/HZSM-5 showed a well stability for the catalyzed
conversion.
Since cellobiose molecule is comprised of two glucose
monomers via 1,4-β-glycoside bond, the synthesis of sorbitol
from cellobiose has to go through the tandem hydrolysis and
hydrogenation processes. Hence, bifunctional catalysts and
catalytic systems were continuously developed for this
conversion [12–16]. In 2006, Liu et al. [12] found that Ru
nanoparticles dispersed in an acidic aqueous medium (pH
2 Experimental
2
.1 Materials
Cellobiose, glucose, and sorbitol were purchased from J&K
Scientific Ltd. (China). Ni(NO ) ·6H O and Co(NO ) ·6H O
3
2
2
3 2
2
were purchased from Sinopharm Chemical Reagent Co. Ltd.
(China). HZSM-5 zeolite (Si/Al=25), HY, and MOR were
purchased from Nankai University Catalyst Co., Ltd. (Chi-
na). Cellobitol was prepared by using previously reported
method [17] and analyzed by using liquid chromatography
mass spectrometry (LC-MS) and high performance liquid
chromatography (HPLC).
2
.0) could efficiently catalyzed converting the cellobiose into
sorbitol. Noteworthily, the authors found that acidic reaction
environment was a necessary condition. New carbon-based
materials, such as carbon-nanotubes (CNTs) and modified
activated carbon have been widely applied in the catalyst
fabrications, since their high hydrothermal stability and
surface paintability [13]. Wang et al. [4] reported the acidic
groups modified CNTs supported Ru nanoparticles catalyst
for the conversion of cellobiose. 87% yield of sorbitol was
2
.2 Catalyst preparation
obtained at 150 °C and 2 MPa H within 30 min. Cellobitol
2
HZSM-5 supported catalysts were prepared via a step im-
pregnation method. 5.0 g HZSM-5 zeolite was impregnated
with 1.45 g Co(NO ) . The mixture was stirred for 12 h at
was confirmed as the key intermediate in this transformation.
Heeres et al. [11] developed mesoporous carbon supported
Ru catalyst (Ru/CMK-3) for the conversion of cellobiose.
3
2
2
5 °C and dried at 105 °C over night. The dried sample were
9
1.1% yield of sorbitol was given at 180 °C and 5 MPa H2
re-dissolved with 2.91 g Ni(NO ) in water and stirred for
3
2
over Ru/CMK-3 catalyst in aqueous phase. The excellent
catalytic activity was due to abundant strong acid sites in the
Ru/CMK-3 catalyst. Recently, Lopez-Sanchez et al. [14]
synthesized a bi-functional Ru catalyst with strong acidic
resin A-15 (Amberlyst-15) for the same conversion. 81%
yield of sorbitol was formed over 3% RuNps/A15 within 5 h.
Pereira et al. [15] discovered that introducing Ni into the Ru
based catalysts would obviously improve the catalytic ac-
tivity. Similarly, Alonso et al. [16] found that the catalytic
activity of Ni/MCM-48 was remarkably improved with in-
troducing trace Ru metal. However, non-noble metal based
catalysts were barely reported for the conversion of cello-
biose. Considering the high cost of noble metal based cata-
lysts, non-noble metal based catalysts are more attractive for
the large-scale application.
another 12 h under room temperature. After that, the water
was evaporated from the solution and the solids were dried at
1
05 °C. The catalysts were calcined at 400 °C in N and
2
reduced at 500 °C in the presence of N /H as reported in our
2
2
previous work [18]. The loadings of Ni and Co were re-
spectively 10% and 5%. Other supported catalysts were also
2
.3 Conversion of glucose and cellobiose
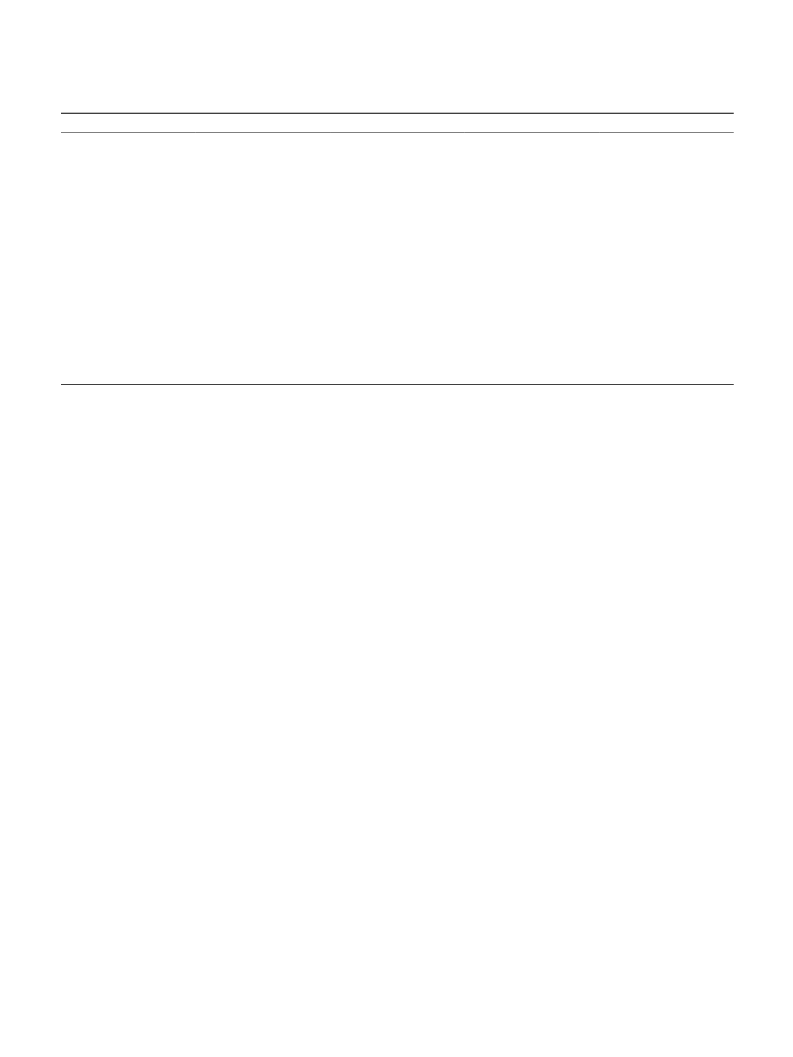
The catalyzed conversions of glucose and cellobiose were
carried out in 25 mL Parr reactor equipped with a manganic
stirrer. In a typical experiment, a solution containing sub-
strate (1 M glucose or 0.5 M cellobiose), 100 mg catalyst and
12 mL deionised water was injected into the reactor. The
In this work, various HZSM-5 supported non-precious
metal based catalysts (i.e. NiCo/HZSM-5, Ni/HZSM-5, and
Co/HZSM-5) were prepared through step impregnation
method and tested for the synthesis of sorbitol. The reaction
reactor was flushed five times with H to remove air and
2
temperature and H pressure both showed an obvious impact
maintained at specifies hydrogen pressure (1–6 MPa). The
sealed reactor was heated to desired temperature with a
heating rate of 10 °C/min at 1000 r/min. The reaction tem-
perature was maintained at a certain value, i.e. in a range of
100 to 200 °C for 1–6 h. After the reaction, the reactor was
cooled to room temperature and the product mixture was
collected and analyzed by HPLC.
2
on the catalytic performance and the highest catalytic activity
was observed over the NiCo/HZSM-5 catalyst. The forma-
tion of NiCo alloy was confirmed by X-ray diffraction
(XRD), X-ray photoelectron spectroscopy (XPS), energy
dispersive X-ray spectroscopy (EDX) and transmission
electron microscopy (TEM) characterizations. Compared

 Zada, Bakht
Zada, Bakht
 Yan, Long
Yan, Long
 Fu, Yao
Fu, Yao