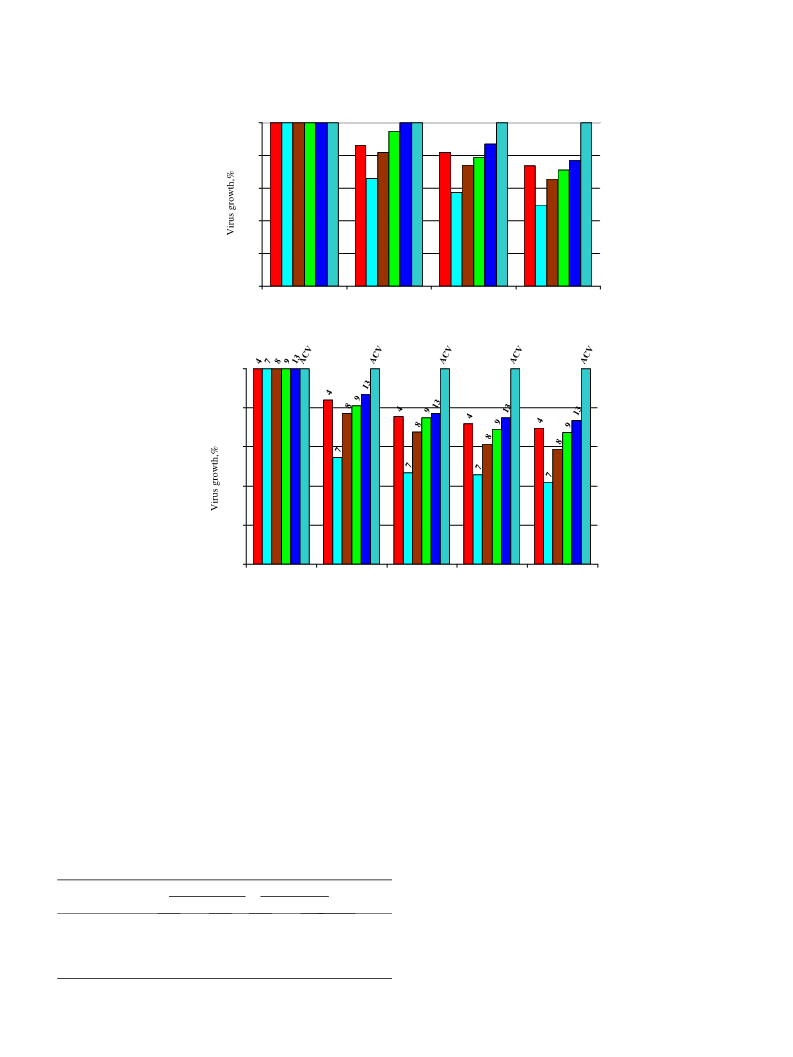
N. F. Zakirova et al. / Bioorg. Med. Chem. 20 (2012) 5802–5809
5807
400 MHz): d 7.78 (s, 1H, H-8), 5.37 (s, 2H, H-10), 3.86–3.88 (m, 2H,
H-30), 3.71–3.73 (m, 2H, H-20), 3.43–3.45 (m, 8H, CH2OCH2), 2.77–
2.79 (m, 8H, CH2NCH2). 31P-NMR (D2O, 162 MHz): d 15.92. 13C-
NMR (D2O, 101 MHz): d 159.67 (C-4), 154.64 (C-2), 152.08 (C-6),
138.66 (C-8), 117.44 (C-5), 73.04 (C-10), 70.60 (d, J = 8.04 Hz, C-
30), 66.44 (d, J = 4.92 Hz, C-20), 64.39 (d, J = 4.01 Hz, CH2OCH2),
45.21 (d, J = 3.86 Hz, CH2NCH2). MS (ESI+) m/z 443,1711 (M).
0.66 mg). Purification and freeze-drying gave 8.8 mg (10%) of (10)
as a white lyophilisate. UV (H2O): kmax 250 nm (9700). 1H-NMR
(D2O, 400 MHz): d 7.79 (s, 1H, H-8), 5.34 (s, 2H, H-10), 3.92–3.96
(m, 2H, H-30), 3.68–3.72 (m, 2H, H-20), 3.22 (t, 2H, J = 6.08 Hz,
(CH3)2NCH2CH2NH), 2.80 (t, 2H, J = 6.08 Hz, (CH3)2NCH2CH2NH),
2.68 (s, 6H, CH3N). 31P-NMR (D2O, 162 MHz): 10.86. 13C-NMR
(D2O, 101 MHz): d 160.94 (C-4), 157.11 (C-2), 153.18 (C-6), 143.10
(C-8), 119.01 (C-5), 74.91 (C-10), 69.35 (d, J = 5.27 Hz, C-30), 65.42
(d, J = 6.99 Hz, C-20), 58.56 (d, J = 4.21 Hz, (CH3)2 NCH2CH2), 45.72
((CH3)N), 42.25 (d, J = 2.51 Hz, (CH3)2NC H2CH2). HRMS (ESIꢀ) calcd
for C12H18N6O5P (M-NH4)ꢀ 374,1342 (M-NH4)ꢀ; found 374,1322.
4.2.7. 9-(2-Hydroxyethoxymethyl)guanine, phosphoromonomo
rpholidate (7)
Product (7) was prepared according to standard procedure A
from ACV (50.0 mg, 0.22 mmol), phosphorus oxychloride (51.0
mg, 0.33 mmol) and morpholine (57.5 mg, 0.66 mmol). Purification
and freeze-drying gave 40 mg (45%) of (7) as a white lyophilisate.
UV (H2O): kmax 250 nm (10200). 1H-NMR (D2O, 400 MHz): d 7.90
(s, 1H, H-8), 5.47 (s, 2H, H-10), 3.83–3.87 (m, 2H, H-30), 3.70–3.73
(m, 2H, H-20), 3.52 (t, 4H, J = 4.40 Hz, CH2OCH2), 2.84 (dt, 4H,
J = 4.40 Hz, J = 4.80 Hz, CH2NCH2). 31P-NMR (D2O, 162 MHz): d
8.18. 13C-NMR (D2O, 101 MHz): d 160.21 (C-4), 155.07 (C-2),
152.94 (C-6), 140.28 (C-8), 118.01 (C-5), 72.81 (C-10), 70.05 (d,
J = 8.21 Hz, C-30), 65.84 (d, J = 5.08 Hz, C-20), 63.51 (d, J = 4.11 Hz,
CH2OCH2), 46.51 (d, J = 3.79 Hz, CH2NCH2). MS(ESI+) m/z 374,1119
(M+H+), 396,0944 (M+Na+), 412,0685 (M+K+); HRMS (ESIꢀ) calcd
for C12H18N6O6P (M-NH4)ꢀ:373,1026 (M-NH4)ꢀ; found 373,0085.
4.2.11. 9-(2-Hydroxyethoxymethyl)guanine, phosphoromono-
3-dimethylamino-1-propylamidate (11)
Product (11) was prepared according to standard procedure A
from ACV (50.0 mg, 0.22 mmol), phosphorus oxychloride (51.0
mg, 0.33 mmol) and 3-dimethylamino-1-propylamine (67.5 mg,
0.66 mmol). Purification and freeze-drying yielded 9.8 mg (11%) of
(11) as a white lyophilisate. UV (H2O): kmax 250 nm (9600). 1H-
NMR (D2O, 400 MHz): d 7.79 (s, 1H, H-8), 5.34 (s, 2H, H-10), 3.92–
3.96 (m, 2H, H-30), 3.68–3.72 (m, 2H, H-20), 3.65-3.69 (m, 2H,
CH2CH2CH2NH), 3.00 (t, 2H, J = 6.78 Hz, (CH3)2NCH2CH2CH2), 2.73
(s, 6H, (CH3)2N), 1.71–1.79 (m, 2H, CH2CH2CH2NH). 31P-NMR (D2O,
162 MHz): d 10.55. 13C-NMR (D2O, 101 MHz): d 162.01 (C-4),
156.83 (C-2), 154.74 (C-6), 142.83 (C-8), 120.01 (C-5), 75.02 (C-10),
71.49 (d, J = 4.76 Hz, C-30), 65.89 (d, J = 8.03 Hz, C-20), 54.61 (d,
J = 4.12 Hz, (CH3)2NCH2CH2CH2), 45.17 ((CH3)2N), 43.24 (d, J =
2.57 Hz, (CH3)2NCH2CH2CH2), 26.97 (d, J = 2.29 Hz, (CH3)2NCH2
CH2CH2). HRMS (ESI-) calcd for C13H20N6O5P (M-NH4)ꢀ 388,1499
(M-NH4)ꢀ; found 388,1465.
4.2.8. 9-(2-Hydroxyethoxymethyl)guanine, phosphoromono-N-
morpholinoethylamidate (8)
Product (8) was prepared according to standard procedure A
from ACV (50.0 mg, 0.22 mmol), phosphorus oxychloride (51.0 mg,
0.33 mmol) and 2-morpholinoethylamine (86.0 mg, 0.66 mmol).
Purification and freeze-drying gave 11.5 mg (12%) of (8) as a white
lyophilisate. UV (H2O): kmax 250 nm (9800). 1H-NMR (D2O,
400 MHz): d 7.79 (s, 1H, H-8), 5.34 (s, 2H, H-10), 3.86–3.89 (m, 2H,
H-30), 3.81 (br s, 4H, CH2OCH2), 3.73–3.75 (m, 2H, H-20), 2.92–2.98
(m, 6H, CH2CH2NH + CH2NCH2), 2.81–2.87 (m, 2H, CH2CH2NH).
31P-NMR (D2O, 162 MHz): d 9.26. 13C-NMR (D2O, 101 MHz): d
159.08 (C-4), 154.39 (C-2), 153.38 (C-6), 140.99 (C-8), 118.14 (C-
5), 74.81 (C-10), 69.34 (d, J = 5.56 Hz, C-30), 66.71 (d, J = 7.64 Hz, C-
20), 66.37 (2s, CH2OCH2), 53.61 (d, J = 2.58 Hz, CH2CH2NH), 53.07
(2s, CH2NCH2), 43.11 (d, J = 4.04 Hz, CH2CH2NH). HRMS (ESIꢀ) calcd
for C14H20N6O6P (M-NH4)ꢀ 416,1448 (M-NH4)ꢀ; found 416,1419.
4.2.12. 9-(2-Hydroxyethoxymethyl)guanine, phosphorodihexyla
midate (12)
Product (12) was prepared according to standard procedure B
from ACV (50.0 mg, 0.22 mmol), phosphorus oxychloride
(51.0 mg, 0.33 mmol) and hexylamine (223 mg, 2.2 mmol). Accord-
ing to NMR data product (12) contained ACV admixture (1) in the
quantity of 30%. UV (H2O): kmax 250 nm (9600). 1H-NMR (D2O,
400 MHz): d 7.93 (s, 1H, H-8), 5.56 (s, 2H, H-10), 3.87–3.91 (m,
2H, H-30), 3.72–3.76 (m, 2H, H-20), 2.46–2.53 (m, 4H, CH2NH),
1.07–1.24 (m, 16H, CH3(CH2)4CH2NH), 0.78 (t, 3H, J = 7.32, CH3).
31P-NMR (D2O, 162 MHz): d 16.05.
4.2.9. 9-(2-Hydroxyethoxymethyl)guanine, phosphoromono-
dipropylamidate (9)
4.2.13. 9-(2-Hydroxyethoxymethyl)guanine, phosphoromonohe
xylamidate (13)
Product (9) was prepared according to standard procedure A
from ACV (50.0 mg, 0.22 mmol), phosphorus oxychloride
(51.0 mg, 0.33 mmol) and dipropylamine (67 mg, 0.66 mmol).
Purification and freeze-drying yielded 36.5 mg (41%) of (9) as a
white lyophilisate. UV (H2O): kmax 249 nm (10000). 1H-NMR
(D2O, 400 MHz): d 7.91 (s, 1H, H-8), 5.48 (s, 2H, H-10), 3.77–3.81
(m, 2H, H-30), 3.71–3.73 (m, 2H, H-20), 2.63–2.70 (m, 4H,
CH2CH2CH3), 1.26–1.36 (m, 4H, CH2CH2CH3), 0.71 (t, 6H,
J = 7.48 Hz, CH2CH2CH3). 31P-NMR (D2O, 162 MHz): d 10.76. 13C-
NMR (D2O, 101 MHz): d 161.67 (C-4), 156.80 (C-2), 154.47 (C-6),
142.78 (C-8), 118.92 (C-5), 75.70 (C-10), 71.88 (d, J = 7.68 Hz, C-
30), 65.89 (d, J = 5.45 Hz, C-20), 50.73 (d, J = 3.43 Hz, CH2CH2CH3),
24.24 (d, J = 2.22 Hz, CH2CH2CH3), 13.45 (CH2CH2CH3). MS (ESI+)
m/z 388,1624 (M-NH3), 410,1443 (M-NH4+Na), 426,1183 (M-
NH4+K); HRMS (ESIꢀ) calcd for C14H24N6O5P (M-NH4)ꢀ: 387,1546
(M-NH4)ꢀ; found 387,1508.
Product (13) was prepared according to standard procedure A
from ACV (50.0 mg, 0.22 mmol), phosphorus oxychloride (51.0
mg, 0.33 mmol) and hexylamine (67.0 mg, 0.66 mmol). Purification
and freeze-drying yielded 15 mg (17%) of (13) as a white lyophili-
sate. UV (H2O): kmax 250 nm (9700). 1H-NMR (D2O, 400 MHz): d
7.92 (s, 1H, H-8), 5.49 (s, 2H, H-10), 3.84–3.89 (m, 2H, H-30), 3.71–
3.74 (m, 2H, H-20), 2.56–2.62 (m, 2H, CH2NH), 1.22–1.29 (m, 4H,
CH3(CH2)2CH2CH2CH2NH), 1.07–1.13 (m, 4H, CH3CH2CH2(CH2)3NH),
0.77 (t, 3H, J = 7.32 Hz, CH3). 31P-NMR (D2O, 162 MHz): d 8.56. 13C-
NMR (D2O, 101 MHz): d 164.08 (C-4), 156.37 (C-2), 155.01 (C-6),
141.14 (C-8), 119.92 (C-5), 74.78 (C-10), 69.88 (d, J = 5.11 Hz, C-30),
65.03 (d, J = 8.04 Hz, C-20), 45.17 (d, J = 3.33 Hz, CH2(CH2)4CH3),
31.75 ((CH2)3CH2CH2CH3), 28.82 (d, J = 2.2 Hz, CH2CH2(CH2)3CH3),
25.79 (d, J = 2.5 Hz, (CH2)2CH2(CH2)2CH3), 22.37 ((CH2)4CH2CH3),
14.14 (CH3). HRMS (ESIꢀ) calcd for C14H24N6O5P (M-NH4)ꢀ:
387,1546 (M-NH4)ꢀ; found 387,1578.
4.2.10. 9-(2-Hydroxyethoxymethyl)guanine, phosphoromono-
N,N-dimethylaminoethylamidate (10)
4.3. Chemical stability
Product (10) was prepared according to standard procedure A
from ACV (50.0 mg, 0.22 mmol), phosphorus oxychloride
(51.0 mg, 0.33 mmol) and dimethylaminoethylamine (58 mg,
Hydrolysis of the compounds (3-13) was performed in acidic
(pH 2, KCl and HCl, 25 ml of 0.2 M KCl and 6.5 ml of 0.2 M HCl

 Zakirova, Natalia F.
Zakirova, Natalia F.
 Shipitsyn, Alexander V.
Shipitsyn, Alexander V.
 Jasko, Maxim V.
Jasko, Maxim V.
 Prokofjeva, Maria M.
Prokofjeva, Maria M.
 Andronova, Valeria L.
Andronova, Valeria L.
 Galegov, Georgiy A.
Galegov, Georgiy A.
 Prassolov, Vladimir S.
Prassolov, Vladimir S.
 Kochetkov, Sergey N.
Kochetkov, Sergey N.