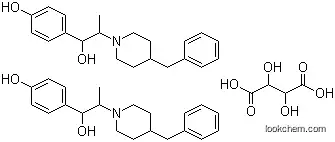
23210-58-4 Usage
Description
Ifenprodil Hemihydrate is a phenylethanolamine derivative that acts as a vasodilator and adrenergic antagonist. It possesses anticonvulsant and antinociceptive properties, making it a valuable compound for treating various medical conditions. Ifenprodil Hemihydrate stimulates blood circulation and is known to block G protein-coupled inwardly-rectifying potassium channels (GIRKs) while interacting with α1 adrenergic, N-methyl-D-aspartate (NMDA), and serotonin receptors. It is a crystalline solid in its chemical form.
Uses
Used in Pharmaceutical Industry:
Ifenprodil Hemihydrate is used as a vasodilator for treating cerebrovascular diseases and peripheral arterial obliterative disease. Its vasodilating properties help improve blood flow to the affected areas, providing relief from the symptoms associated with these conditions.
Used in Neurological Applications:
Ifenprodil Hemihydrate is used as an anticonvulsant and antinociceptive agent for treating posttraumatic stress disorder and other neurological disorders. Its ability to block NMDA receptors and interact with serotonin receptors makes it a promising candidate for managing these conditions.
Used in Research and Development:
Ifenprodil (+)-tartrate salt has been used in various research studies to test its action in KCl-induced cortical spreading depression (CSD) in animals. It is also used to study its effects on electrophysiological properties and spontaneous spikes in neocortical pyramidal cells, as well as to investigate its therapeutic effects on different conditions.
Therapeutic Function
Vasodilator
Biological Activity
Ifenprodil hemitartrate is a NMDA receptor antagonist, acting at the polyamine site. Also an α-adrenergic vasodilator. σ2 ligand displaying about 3-fold selectivity over σ1 sites.
Biochem/physiol Actions
NMDA antagonist acting at the polyamine site; neuroprotective agent; α-adrenergic central and peripheral vasodilator; α2 adrenergic receptor ligand.
in vitro
Ifenprodil treatment of oocytes, at NR1A/NR2B and NR1A/NR2A receptors, inhibited NMDA-induced currents with IC50 of 0.34 μM and 146 μM, respectively, and voltage-clamped at -70 mV. Ifenprodil acts as a weak open channel blocker of NR1A/NR2A receptors, the degree of inhibition of NR1A/NR2A receptors by 100 μM fenprodil is not affected by extracellular glycine concentration. Increasing glycine concentration decreased the inhibitory effect of 1mMIfenprodil on NR1A/NR2B receptors. Ifenprodil (10 μM) acts on young rat cortical neurons, a single population of receptors with high affinity for glycine, inhibits almost all NMDA receptor-evoked currents. Ifenprodil (10 μM) inhibited a significant proportion of high- and low-affinity components in aged rat cortical neurons, revealing three pharmacologically distinct distribution populations of NMDA receptors in a single neuron. Ifenprodil antagonizes NMDA receptors in an activity-dependent manner and also enhances the receptor's affinity for glutamate recognition site agonists. In the inhibition curves of Ifenprodil on 10μM and 100μM NMDA-evoked currents, IC50 was 0.88μM and 0.17μM, respectively. Ifenprodil (3μM) can enhance the control level of cultured rat cortical neurons. Enhanced by about 200%. Ifenprodil has a 39- and 50-fold enhanced affinity for NMDA receptors in the agonist-bound activated and desensitized states compared to the resting, agonist-unbound state. Ifenprodil binds to NMDA receptors with a 6-fold increased affinity for glutamate receptor agonists.
Check Digit Verification of cas no
The CAS Registry Mumber 23210-58-4 includes 8 digits separated into 3 groups by hyphens. The first part of the number,starting from the left, has 5 digits, 2,3,2,1 and 0 respectively; the second part has 2 digits, 5 and 8 respectively.
Calculate Digit Verification of CAS Registry Number 23210-58:
(7*2)+(6*3)+(5*2)+(4*1)+(3*0)+(2*5)+(1*8)=64
64 % 10 = 4
So 23210-58-4 is a valid CAS Registry Number.
InChI:InChI=1/2C21H27NO2.C4H6O6/c2*1-16(21(24)19-7-9-20(23)10-8-19)22-13-11-18(12-14-22)15-17-5-3-2-4-6-17;5-1(3(7)8)2(6)4(9)10/h2*2-10,16,18,21,23-24H,11-15H2,1H3;1-2,5-6H,(H,7,8)(H,9,10)/t;;1-,2-/m..1/s1