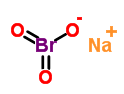
Quick Details
- ProName: Sodium bromate
- CasNo: 7789-38-0
- Molecular Formula: NaBrO3
- Appearance: Solid
- Application: Industrial etc.
- Purity: ≥99%
- Transportation: containers
- LimitNum: 1 Metric Ton
Superiority
Sodium bromate Basic information
Synonyms: bromatedesodium;bromatedesodium[french];dyetone;neutralizerk-126;neutralizerk-140;neutralizerk-938;BROMI…
Details
Sodium bromate Basic information
Synonyms: bromatedesodium;bromatedesodium[french];dyetone;neutralizerk-126;neutralizerk-140;neutralizerk-938;BROMIDE ION CHROMATOGRAPHY STANDARD;BROMIDE STANDARD
MF: BrNaO3
MW: 150.89
EINECS: 231-830-3
Product Categories: Inorganic Chemicals;Inorganics;Analytical Reagents for General Use;Puriss p.a.;Q-S, Puriss p.a.;Inorganic Salts;Synthetic Reagents
Mol File: 7789-38-0.mol
Sodium bromate Chemical Properties
mp 755 °C(lit.)
bp 1390 °C
density 3.339 g/mL at 25 °C(lit.)
vapor pressure 1 mm Hg ( 806 °C)
refractive index 1.594
solubility H2O: 1 M at 20 °C, clear, colorless
Water Solubility 364 g/L (20 oC)
Merck 14,8593
Stability: Stable. Oxidizer. Incompatible with finely powdered metals, alcohols, strong acids, strong reducing agents.
CAS DataBase Reference 7789-38-0(CAS DataBase Reference)
EPA Substance Registry System Bromic acid, sodium salt(7789-38-0)
Sodium bromate Usage And Synthesis
Chemical Properties white crystals
General Description A white crystalline solid. May explode under prolonged exposure to heat or fire. Used in chemical analysis.
Air & Water Reactions Water soluble.
Reactivity Profile Sodium bromate is an oxidizing agent. May react violently with combustibles and reducing agents. Reacts with textiles, oil, fat, grease, sugar, sawdust and ammonium salts, carbon, phosphorus, metal powders and sulfides with hazard of fire and explosion [Handling Chemicals Safely 1980 p. 831]. A mixture of finely divided aluminum with finely divided Sodium bromate explodes by heat, percussion, and friction [Mellor 2:310 1946-47].
Health Hazard Toxic by ingestion. Inhalation of dust is toxic. Fire may produce irritating, corrosive and/or toxic gases. Contact with substance may cause severe burns to skin and eyes. Runoff from fire control or dilution water may cause pollution.
Fire Hazard These substances will accelerate burning when involved in a fire. May explode from heat or contamination. Some may burn rapidly. Some will react explosively with hydrocarbons (fuels). May ignite combustibles (wood, paper, oil, clothing, etc.). Containers may explode when heated. Runoff may create fire or explosion hazard.
Sodium bromate Preparation Products And Raw materials
Preparation Products Bromine-->Potassium bromate -->3-BROMO-6-METHYL-2-OXO-1(2H)-PYRAZINEACETIC ACID ETHYL ESTER-->ethyl 2-(3,4-dihydro-6-methyl-2,3-dioxopyrazin-1(2H)-yl)acetate-->3-Bromoanisole-->4-Aminomethylquinoline hydrochloride-->DI-N-OCTYL SULFOXIDE
Raw materials Sodium hydroxide-->Bromine-->Barium chloride-->CALCIUM BROMATE-->BARIUM BROMATE
No L/C.Payment by or TT.
Pay freight cost and sample cost by to get a sample.
NOTICE!!!
Only Interested buyers can apply.
Thanks