
Journal of Solution Chemistry p. 775 - 784 (1981)
Update date:2022-08-11
Topics:
 Hoeiland, H.
Hoeiland, H.
 Hald, L.H.
Hald, L.H.
 Kvammen, O.J.
Kvammen, O.J.
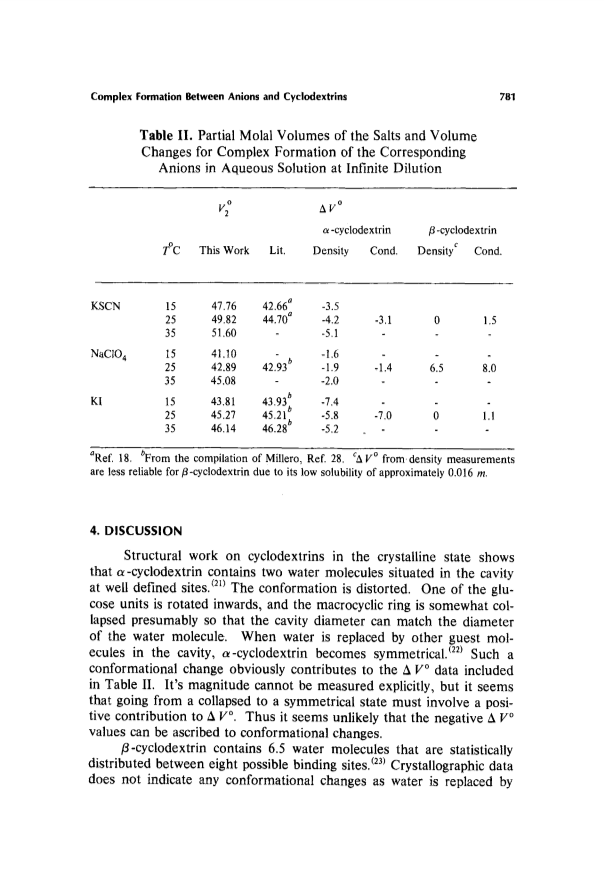
Partial molal volume changes during complex formation between SCN(1-), I(1-), and ClO4(1-) and α- and β-cyclodextrin have been determined by two independent methods of measurements; one based on density measurement and subsequent calculation of apparent molal volumes, the other on differentiating the association constants with respect to pressure.Results from the two methods are in good agreement.Negative volume changes were observed for complex formation between the anions and α-cyclodextrin while zero or slightly positive values were observed for complex formation with β-cyclodextrin.The result is consistent with the idea that the anions do not become dehydrated as they form complexes with cyclodextrins.
View More








website:http://www.afinechem.com
Contact:+86-571-85134551
Address:No. 206 Zhen Hua Road, Hangzhou 310030, Zhejiang, China
Jinhua City Mingzhu Pharmaceutical Co.,Ltd.
Contact:15857995878 0579-82207761
Address:No.169 Shenze Road, New Area,Jinpan Development Zone, Jinhua
Contact:+86-532-80762375
Address:No. 6, Hongkong Middle Road, Qingdao, China
ZHIJIANG ZENVA SINO COMMERCE AND TRADE CO., LTD.
website:http://www.zenvasino.com
Contact:+86-138-72658998
Address:Shibeishan Road,Zhijiang, Hubei, China
Shandong Xiangde Biotechnology Co., Ltd
Contact:+86 -15066639877
Address:Sanba street
Doi:10.1016/j.tetasy.2009.07.048
(2009)Doi:10.1016/j.molcata.2015.05.009
(2015)Doi:10.1021/jo01362a028
(1957)Doi:10.1039/j29710001625
(1971)Doi:10.1021/ja01245a513
(1943)Doi:10.1021/jm00333a045
(1964)