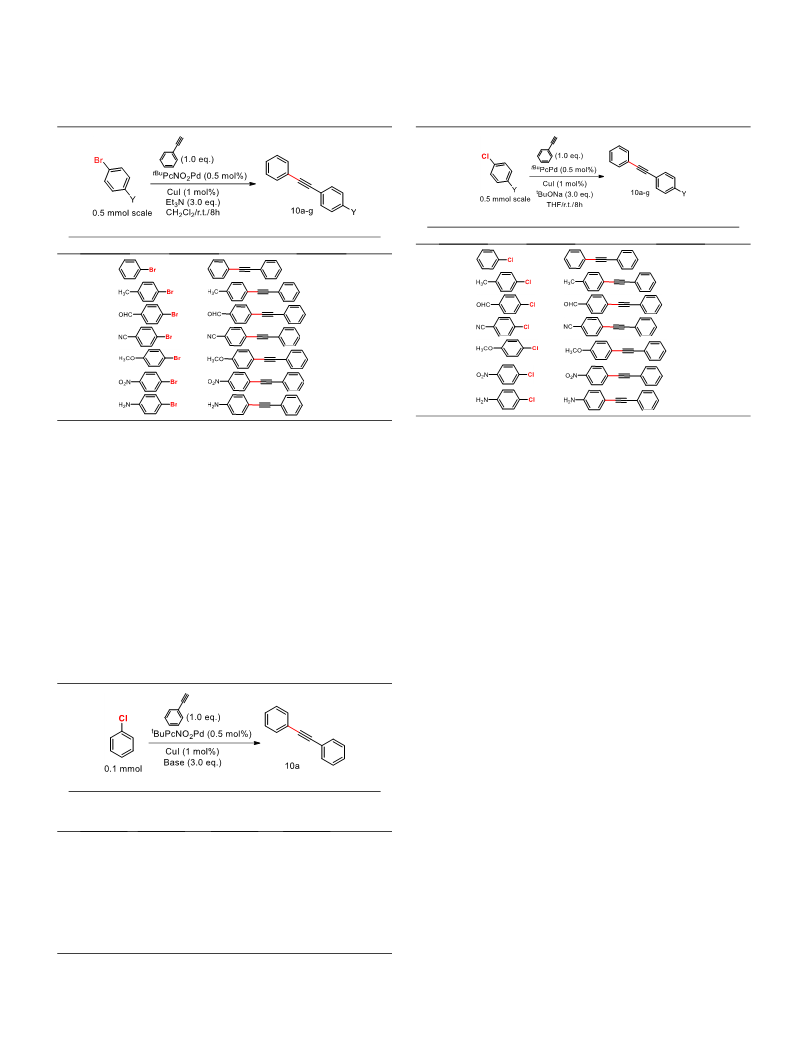Y.B. Platonova et al. / Journal of Catalysis 391 (2020) 224–228
225
Scheme 1. C-alkynylation with terminal acetylenes under palladium catalysis and Sonogashira coupling conditions.
unsymmetrical A3B-type phthalocyanines containing electron
withdrawing groups (F, Cl, NO2, CN) for determination of their
influence on catalytic activity of complexes. The choice of these
complexes as ligands is associated with high solubility in various
organic solvents and stability in solutions in wide range of temper-
atures. It should also be noted that an additional reason for obtain-
ing palladium(II) A3B-type phthalocyanines was very poor
solubility of tetra-substituted analogs in most organic solvents
even at heating.
These complexes are good catalytic model systems for use in
cross-coupling reaction, because they don’t catalyze process in
precatalytic condition and «homeopathic release» of high active
palladium species to prevent formation of palladium black during
the reaction. It also should be noted that palladium complexes
with macrocyclic ligands such as phthalocyanines and porphyri-
nes posess good solubility and chemical stability in wide range
of temperatures that is very favor for use in different catalytic
processes.
4-fluorophthalonitrile or 4-chlorophthalonitrile respectively in
conditions of template synthesis (Scheme 3) with 20–25% yields.
It should be noted that low yields of palladium complexes of
A3B-type phthalocyanines is associated with limitation of this
method because of statistically formation of products mixture
(A4, A3B, A2B2). 4-Nitrophthalonitrile was synthesized by nitration
reaction of phtalimide with fuming nitric acid in sulfuric acid and
following two-step formation of nitrile group (Scheme 3) [36]. 4-
Chloro- and 4-cyanophthalonitrile were obtained via diazotiation
of 4-aminophthalonitrile with good yields by known literature
methods (Scheme 3) [37]. We used Balz-Schiemann reaction for
synthesis of 4-fluorophthalonitrile, which is based on formation
of diazonium tetrafluoroborate salt through diazotiation reaction
of 4-aminophthalonitrile with nitrosonium tetrafluoroborate with
following it’s decomposition at heating in 1,2-dichlorobenzene
with 70% yield (Scheme 3) [38].
The formation of palladium complexes was confirmed by UV–
Vis spectroscopy, ESI-HRMS mass spectrometry and 1H NMR spec-
troscopy. UV–Vis spectrum of complexes 1 and 6–9 showed non-
splitting Q and Soret band that is typically for phthalocyanine com-
plexes. It should be noted that 1H NMR spectra of phthalocyanine
palladium complexes are complicated because they consists of
mixture of regioisomers which are not separated by chromatogra-
phy and HPLC. We observed proton signals of tert-butyl groups and
aromatic phenyl rings in 1H NMR spectrum.
The palladium complex of tetra-tert-butyl-substituted phthalo-
cyanine 1 was prepared via one-step protocol based on metal-
catalyzed template synthesis from easy available
4-tert-butyl phthalonitrile (Scheme 2). The synthesis of com-
plex 1 was carried out by using of excess of bis(benzonitrile)palla
dium(II) dichloride and DBU as base at heating in anhydrous ben-
zonitrile during 12 h. The control of reaction was monitored by TLC
(silica, dichloromethane) and UV–Vis spectroscopy. After comple-
tion of the reaction, reaction mixture was cooled, solvent was
evaporated in vacuo and target complex 1 was isolated by column
chromatography on silica.
In case of unsymmetrical A3B-type phthalocyanines containing
mono electron withdrawing group, target palladium complexes
were prepared by mixed condensation
4-tert-butylphthalonitrile with 4-nitrophthalonitrile, 4-cyano-
phthalonitrile,
The m/z values in the ESI-HRMS spectra confirm the structure of
the complex and the isotopic distribution which is typical for such
palladium complexes is in accordance with the calculated data.
The potential advantages of the novel catalyst system based on
palladium complexes of phthalocyanines, the ease of separation
and facile recycling of the catalyst and the absence of metal con-
tamination in the product, suggest that an efficient Sonogashira
coupling protocol based on the use of this catalyst system and uti-
lizing aromatic halides other than iodides would be of major inter-
est for both industrial and academic applications.
Herein, we report our results on the development of new cat-
alytic systems based on palladium phthalocyanines and use in
Sonogashira cross-coupling reaction of phenylacetylene with vari-
ous p-substituted aryl bromides. First, we have investigated Pd/Cu-
catalyzed Sonogashira reaction of bromobenzene with pheny-
lacetylene as model substrates under various conditions. The reac-
tions were performed in the presence of tBuPcPd catalysts, CuI as a
co-catalyst, Et3N as base and various solvents in test tubes of Rad-
leys Carousel 6 plus at 25 °C and yields of products were deter-
mined by GC/MS. The reaction conditions explored are
summarized in Table 1.
The first task of our investigation was to estimate of the influ-
ence of different reaction parameters on the catalytic activity in
order to optimize the protocol. The limitations of this protocol have
been tested in respect to catalyst, solvent and reaction time. Bases
Scheme 2. Synthetic pathway for preparation of palladium(II) phthalocyanines.

 Platonova, Yana B.
Platonova, Yana B.
 Tomilova, Larisa G.
Tomilova, Larisa G.
 Volov, Alexander N.
Volov, Alexander N.