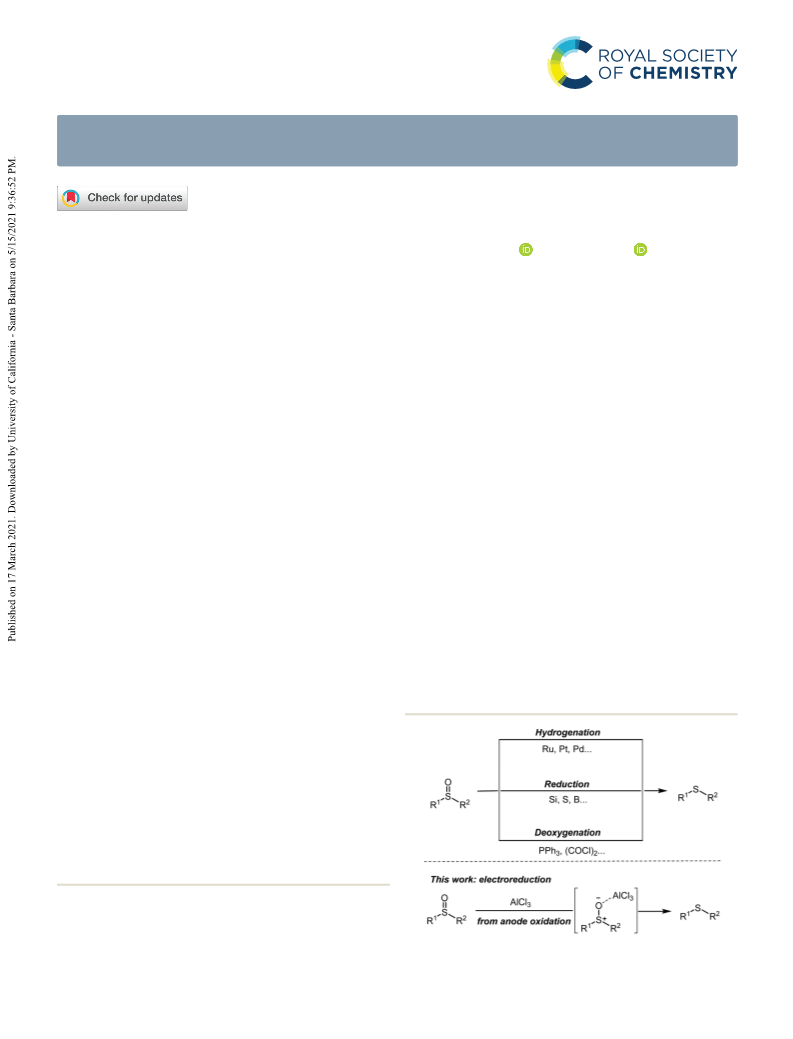
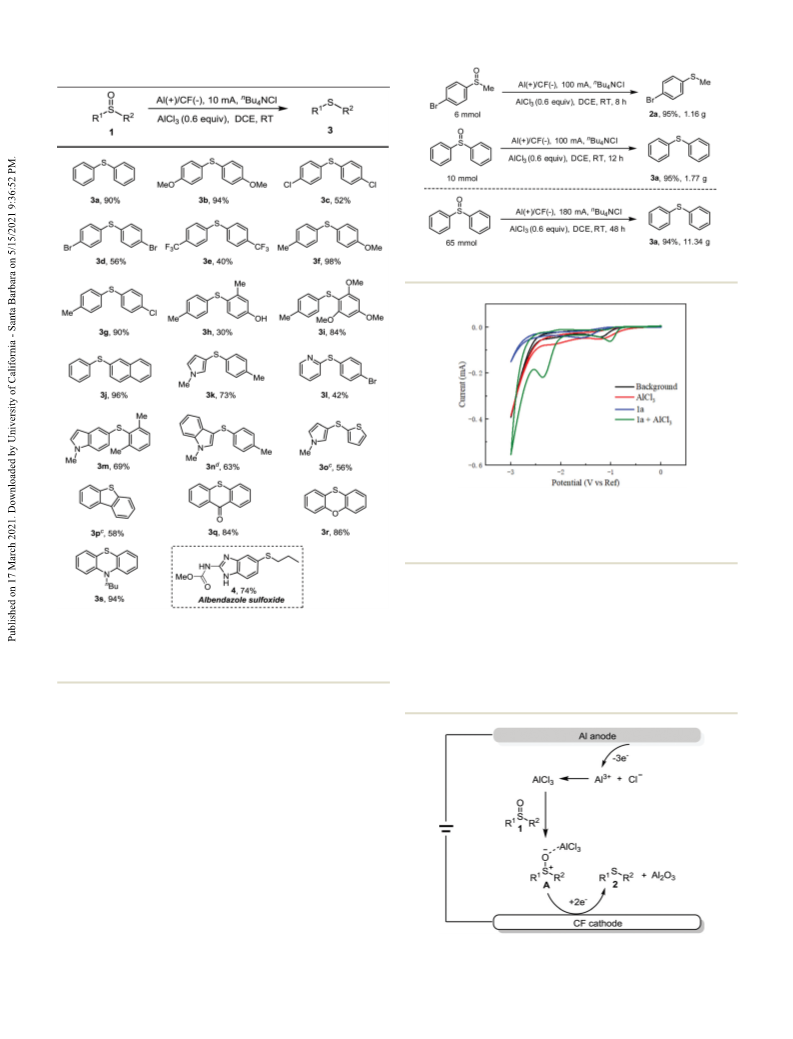
Paper
Green Chemistry
1
a and AlCl
electrolysis and constant potential experiments could further
support the formation of the AlCl –sulfoxide complex (see the
3
would be generated. The results of divided cell
Y. Xiao, Catalysts, 2020, 10, 1339; (d) H. Liu and X. Jiang,
Chem. – Asian J., 2013, 8, 2546–2563; (e) M. Wang, Y. Li and
X. Jiang, Aldrichimica Acta, 2020, 53, 19–25.
3
ESI† for details). Based on the above experiments, the possible
mechanism of the electrochemical process is proposed in
Scheme 3. Initially, AlCl could coordinate with sulfoxide 1 to
3
5 (a) M. Madesclaire, Tetrahedron Lett., 1986, 42, 5459–5495;
(b) V. U. Kukushkin, Coord. Chem. Rev., 1995, 139, 375–407;
(c) H. Firouzabadi and A. Jamalian, J. Sulfur Chem., 2008,
29, 53–97; (d) A.-C. Gaumont, M. Gulea, S. Perrio and
V. Reboul, Compr. Org. Synth. (2nd Ed.), 2014, 8, 535–563;
(e) L. Shiri and M. Kazemi, Res. Chem. Intermed., 2017, 43,
6007–6041; (f) W. Li, X. Chen, T. Zhen, Q. Zou and
W. B. Chen, Chin. J. Org. Chem., 2019, 39, 2443–2457.
generate the Lewis acid–base complex A, which is further
reduced on the cathode by S–O bond cleavage to obtain the
3
+
corresponding sulfide 2. The Al produced from the sacrificial
Al anode could combine with the chloride anion to regenerate
the AlCl3.
6
(a) T. Mitsudome, Y. Takahashi, T. Mizugaki, K. Jitsukawa
and K. Kaneda, Angew. Chem., Int. Ed., 2014, 53, 8348–8351;
(b) A. S. Touchy, H. Siddiki, W. Onodera, K. Kon and
K. Shimizu, Green Chem., 2016, 18, 2554–2560;
Conclusions
In conclusion, we have developed a green and scalable electro-
chemical protocol for the reduction of sulfoxides to sulfides
under mild conditions. The reaction proceeds well using a sub-
(
c) Y. Kuwahara, Y. Yoshimura, K. Haematsu and
H. Yamashita, J. Am. Chem. Soc., 2018, 140, 9203–9210;
d) A. Gevorgyan, S. Mkrtchyan, T. Grigoryan and
V. O. Iaroshenko, ChemPlusChem, 2018, 83, 375–382;
e) K. Yao, Z. Yuan, S. Jin, B. Liu and Z. Zhang, Green
(
3
stoichiometric amount of the Lewis acid AlCl . The Lewis acid
could be regenerated by a combination of chloride anions with
(
3
+
Al waste produced by the oxidation of the sacrificial Al
anode. This method also features a broad substrate scope and
is easily scaled-up. Further application of electrochemical
reduction is currently under investigation.
Chem., 2020, 22, 39–43; (f) S. Fujita, S. Yamaguchi,
S. Yamazoe, J. Yamasaki, T. Mizugaki and T. Mitsudome,
Org. Biomol. Chem., 2020, 18, 8827–8833.
(a) S. Enthaler, Catal. Sci. Technol., 2011, 1, 104–110;
(
7
8
b) F. Ding, J. Jiang, S. Gan and L. Shi, Eur. J. Org. Chem.,
017, 3427–3430; (c) D. Porwal and M. Oestreich, Synthesis,
017, 4698–4702.
2
2
Conflicts of interest
(a) D. J. Harrison, N. C. Tam, C. M. Vogels, R. F. Langler,
R. T. Baker, A. Decken and S. A. Westcott, Tetrahedron Lett.,
There are no conflicts to declare.
2
004, 45, 8493–8496; (b) G. Wang, H. Zhang, J. Zhao,
C. Zhu and S. Li, Angew. Chem., Int. Ed., 2016, 55, 5985–
989; (c) F. Takahashi, K. Nogi and H. Yorimitsu,
Acknowledgements
5
Eur. J. Org. Chem., 2020, 3009–3012.
(a) M. Abbasi, M. R. Mohammadizadeh and Z. Moradi,
Tetrahedron Lett., 2015, 56, 6610–6613; (b) N. Garcia,
This work was financially supported by the National Natural
Science Foundation of China (21572110) and the Natural
Science Foundation of Shandong Province (ZR2020MB005 and
ZR2019MB010).
9
M.
M. R. Pedrosa, F. J. Arnaiz and R. Sanz, RSC Adv., 2016, 6,
7083–27086; (c) A. Zupanc and M. Jereb, Green Chem. Lett.
Rev., 2020, 13, 341–348.
A.
Fernandez-Rodrıguez,
P.
Garcia-Garcia,
2
Notes and references
1
0 (a) Y. Jang, K. T. Kim and H. B. Jeon, J. Org. Chem., 2013,
78, 6328–6331; (b) P. Acosta-Guzman, C. Mahecha-
Mahecha and D. Gamba-Sanchez, Chem. – Eur. J., 2020, 26,
10348–10354; (c) A. K. Clarke, A. Parkin, R. Taylor,
W. P. Unsworth and J. A. Rossi-Ashton, ACS Catal., 2020,
10, 5814–5820.
1
N. Wang, P. Saidhareddy and X. Jiang, Nat. Prod. Rep., 2020,
7, 246–275.
3
2
(a) F. I. Zuniga, D. Loi, K. H. J. Ling and D. D-S. Tang-Liu,
Expert Opin. Drug Metab. Toxicol., 2012, 8, 467–485;
(
b) E. A. Ilardi, E. Vitaku and J. T. Njardarson, J. Med.
Chem., 2014, 57, 2832–2842; (c) M. Feng, B. Tang, S. Liang 11 (a) M. Yan, Y. Kawamata and P. S. Baran, Chem. Rev., 2017,
and X. Jiang, Curr. Top. Med. Chem., 2016, 16, 1200–1216;
d) C. T. Barce-Ferro and N. L. Campos-Domingues, Top.
Med. Chem., 2020, 17, 192–210.
X. Li, W. Ma, H. Li, Q. Zhang and H. Liu, Coord. Chem.
Rev., 2020, 408, 213191.
(a) G. M. F. Batista, P. P. Castro, J. A. Santos, T. Skrydstrup
and G. W. Amarante, Org. Chem. Front., 2021, 8, 326–368;
117, 13230–13319; (b) Y. Jiang, K. Xu and C. Zeng, Chem.
Rev., 2018, 118, 4485–4540; (c) P. Xiong and H.-C. Xu, Acc.
Chem. Res., 2019, 52, 3339–3350; (d) Y. Yuan and A. Lei,
Acc. Chem. Res., 2019, 52, 3309–3324; (e) J. C. Siu, N. Fu and
S. Lin, Acc. Chem. Res., 2020, 53, 547–560; (f) J. L. Rockl,
D. Pollok, R. Franke and S. R. Waldvogel, Acc. Chem. Res.,
2020, 53, 45–61.
(
3
4
(
b) E. M. McGarrigle, E. L. Myers, O. Illa, M. A. Shaw, 12 (a) N. Fu, L. Song, J. Liu, J. Siu and S. Lin, J. Am. Chem.
S. L. Riches and V. K. Aggarwal, Chem. Rev., 2007, 107,
841–5883; (c) R. Zhang, H. Ding, X. Pu, Z. Qian and
Soc., 2019, 141, 14480–14485; (b) P.-F. Zhong, H.-M. Lin,
L.-W. Wang, Z.-Y. Mo, X.-J. Meng, H.-T. Tang and Y.-M. Pan,
5
2776 | Green Chem., 2021, 23, 2773–2777
This journal is © The Royal Society of Chemistry 2021

 Kong, Zhenshuo
Kong, Zhenshuo
 Pan, Chao
Pan, Chao
 Li, Ming
Li, Ming
 Wen, Lirong
Wen, Lirong
 Guo, Weisi
Guo, Weisi