1
68
D. Sun et al. / Applied Catalysis A: General 526 (2016) 164–171
Table 3
Dehydration of 1,2-ethanediol over WO3/SiO2-320 at different reaction temperatures with a feed of 20 wt.% aqueous 1,2-ethanediol solution .
a
Temperature
/o
Conversionb
/%
Selectivity/%b
Ethanal
Coke content
/wt.%
C
1,4-Dioxane
MDX
DEG
TEG
250
280
310
340
17.9
76.0
99.5
99.9
30.6
76.2
92.1
94.8
44.8
18.8
6.5
15.8
3.3
0.1
7.7
0.3
0.0
0.0
0.0
0.0
0.0
0.0
2.6
2.6
3.1
3.8
0.0
2.3
a
Reaction conditions: feed rate, 1.32 cm3
Average activity in the initial 5 h. (MDX, 2-methyl-1,3-dioxolane; DEG, diethylene glycol; TEG, triethylene glycol).
−1
(4.3 mmol h−1); catalyst weight, 0.5 g; H2 flow rate, 30 cm3 min−1
h
.
b
was the intramolecular dehydration product of EG, and the other
products were generated from at least two molecules. At high con-
content in the used catalysts increased with increasing the con-
centration of PeDO. Table 6 shows the dehydration of PeDO over
WO /SiO -320 at different reaction temperatures with a feed of
centrations of H O, the reduced partial pressure of EG and the
2
3
2
products could decrease the possibility of the contact between
two organic molecules, therefore, decrease the selectivity to the
intermolecular dehydration products as well as MDX. Thus, high
pure PeDO. In a similar way of the dehydration of BDO, the conver-
sion of PeDO, the selectivity to 1,3-pentadiene and the coke content
in the used catalysts increased with increasing the temperature,
while the selectivity to BPDX decreased. The maximum selectivity
to pentanal was achieved at 75.1% with a complete conversion at
concentrations of H O were preferable for the formation of ethanal.
2
In a similar way, high concentrations of H O would inhibit the
2
◦
formation of coke because coke is generally formed via polymer-
310 C.
ization of monomer products. Thus, high concentrations of H O are
In the dehydration of BDO and PeDO over WO /SiO -320, the
2
3 2
proposed to be effective for inhibiting the catalytic deactivation.
addition of H O and high temperatures inhibit the formation of
2
acetal, which has the same causes as has been described in the
dehydration of EG (Section 3.2). The additive H O significantly
2
3
.3. Dehydration of 1,2-butanediol and 1,2-pentanediol over
affected the selectivity to dienes. Since the existence of H O poi-
2
WO /SiO -320 catalyst
3
2
soned the Lewis acid sites and the formation of dienes preferred low
H O concentrations, it is supposed that Lewis acid sites involve the
2
Table 4 summarizes the dehydration of BDO over WO /SiO -320
at different reaction temperatures. Butanal, 1-butanol, 2-buten-
-ol, 2-propyl-4-ethyl-1,3-dioxolane (PEDX) and butadiene were
3
2
formation of dienes. In particular, in contrast to the dehydration
of BDO and PeDO to aldehydes over Brønsted acid sites, Lewis acid
sites would catalyze the dehydration of BDO and PeDO to the cor-
responding unsaturated alcohols, 2-buten-1-ol and 2-penten-1-ol,
which could be further converted into dienes.
1
generated as the main products. 2-Buten-1-ol was produced by the
dehydration of the secondary OH with the H at position 3 of BDO.
Hydrogenation of 2-buten-1-ol formed 1-butanol and further dehy-
dration of 2-buten-1-ol formed butadiene. PEDX was generated by
the cyclodehydration of the produced butanal with another BDO.
At a feed of 20 wt.% aqueous BDO solution, a high butanal selectiv-
3.4. Comparison of the dehydration of different 1,2-alkanediols at
different reaction conditions
◦
ity of 93.9% with a complete conversion was achieved at 250 C. At
a feed of pure BDO, however, the selectivity to butanal and the con-
version of BDO decreased to 88.8 and 66.9%, respectively. Because
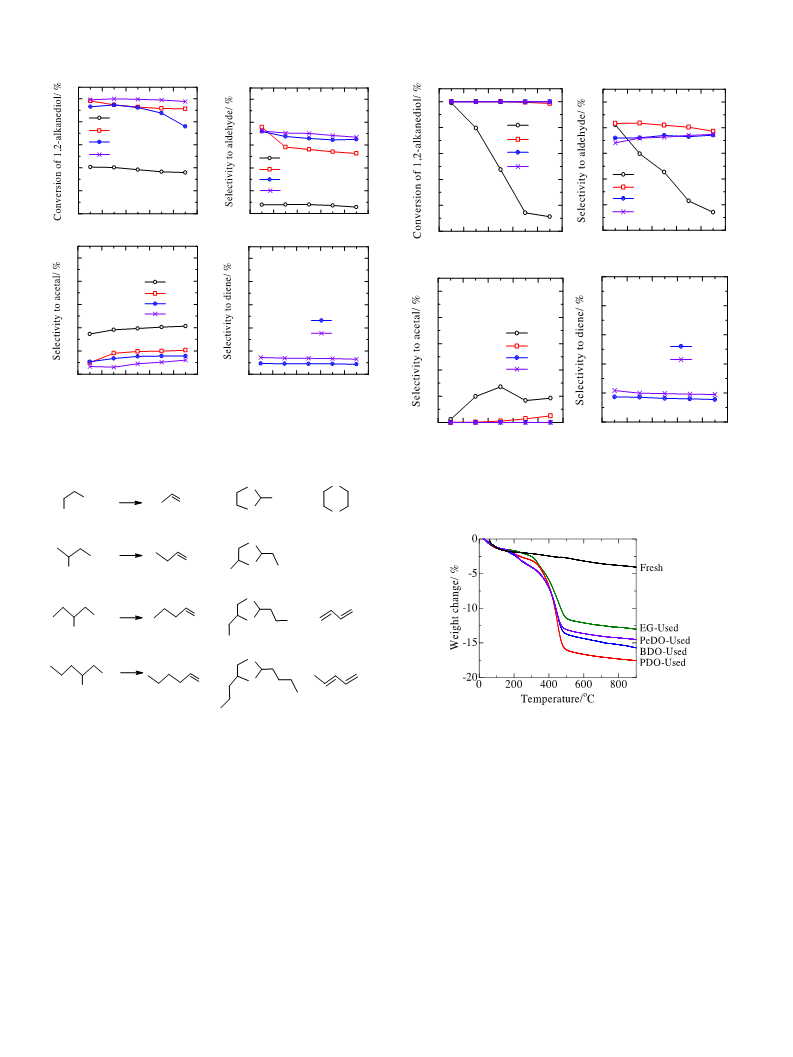
Fig. 4 compares the dehydration of different 1,2-alkanediols
at 250 C with a feed of 20 wt.% aqueous 1,2-alkanediol solu-
◦
H O poisons Lewis acid sites, the high selectivity of butanal at a
tion. Under such reaction conditions, the conversion of each
1,2-alkanediol over WO /SiO -320 was stable (Fig. 4a). However,
2
high concentration of H O suggests that Brønsted acid sites pri-
2
3
2
marily catalyze the dehydration of BDO to butanal. In contrast to
the case in the dehydration of EG, the conversion of BDO monoton-
ically increased with increasing the temperature, and significant
deactivation was not observed even at a feed of pure BDO. The
selectivity to PEDX decreased with increasing the reaction temper-
ature, while the selectivity to butadiene increased. At a feed of pure
in contrast to the complete conversion of PDO, BDO and PeDO,
the conversion of EG was low. This indicates that EG is less reac-
tive than the other 1,2-alkanediols. The dehydration of EG gave
a low selectivity to aldehyde (Fig. 4b), and a high selectivity to
acetal comparing with those in the dehydration of PDO, BDO and
PeDO (Fig. 4c). This indicates that the selectivity in the dehydra-
tion of EG is different from those in the dehydration of the other
1,2-alkanediols, which is probably caused by the lack of an alkyl
group in EG. In another word, the existence of an alkyl group would
inhibit the formation of acetal as well as the etherification of 1,2-
alkanediol, therefore, improve the formation of aldehyde.
◦
BDO, the selectivity to butanal was maximized at 74.9% at 310 C.
The formation of coke on the catalyst surface was also observed in
the dehydration of BDO, and the coke content in the used catalysts
increased with increasing the temperature.
Table 5 summarizes the dehydration of PeDO over WO /SiO -
3
2
◦
3
20 at 250 C at a feed of aqueous PeDO solution with different
Fig. 5 compares the dehydration of different 1,2-alkanediols at
◦
concentrations. Pentanal, 1-pentanol, 2-penten-1-ol, 2-butyl-4-
propyl-1,3-dioxolane (BPDX) and 1,3-pentadiene were generated
as the main products. 2-Penten-1-ol was another intramolecular
dehydration product of PeDO besides pentanal. 1-Pentanol and
250 C with a feed of pure 1,2-alkanediols, and Scheme 2 summa-
rizes the distribution of the main products. The conversion of EG
was still lower than those of PDO, BDO and PeDO under the reac-
tion conditions (Fig. 5a). The conversion decreased with time on
stream in all the reactions, while the decrease in the conversion of
BDO was obvious comparing with that of the other 1,2-alkanediols.
The coke contents in the used catalysts after 5 h reactions were 5.4,
5.1, 8.9 and 7.4% in the dehydration of EG, PDO, BDO and PeDO,
respectively. Thus, the rapid decrease in BDO conversion is consid-
ered to be attributed to the large amount of the accumulated coke
on the catalyst surface. Since conjugate dienes are polymerizable
compounds, the generation of butadiene and 1,3-pentadiene is pro-
posed to associate with the relatively heavy coke formation in the
1
,3-pentadiene were generated through the hydrogenation and
the further dehydration of 2-penten-1-ol, respectively. BPDX was
generated by the cyclodehydration of the produced pentanal with
another PeDO. The selectivity to pentanal was 91.1% with 100%
conversion at a feed of 20 wt.% aqueous PeDO solution. Both the
conversion of PeDO and the selectivity to pentanal decreased with
increasing the concentration of PeDO, while the selectivity to BPDX
and 1,3-pentadiene increased. This indicates that high concentra-
tions of H O are preferable for the formation of pentanal. Coke
2

 Sun, Daolai
Sun, Daolai
 Yamada, Yasuhiro
Yamada, Yasuhiro
 Sato, Satoshi
Sato, Satoshi
 Suganuma, Satoshi
Suganuma, Satoshi
 Katada, Naonobu
Katada, Naonobu