3
44ꢀ JOURNALꢀOFꢀCHEMICALꢀRESEARCHꢀ2009
Table 2ꢀ Reductionꢀofꢀtrihydroxybenzeneꢀ(1u–w)a
b
Yield/%c
Runꢀ
ꢀ
Substrateꢀ
ꢀ
Ni–Al/gꢀ
ꢀ
Alkalineꢀsolution /mLꢀ
Time/hꢀ
ꢀ
ꢀ
7
1g
1
2
3
4
5
6
ꢀ
ꢀ
ꢀ
ꢀ
ꢀ
ꢀ
1u
1u
1u
1u
1v
1w
b
5ꢀ
5ꢀ
5ꢀ
5ꢀ
5ꢀ
5ꢀ
1%ꢀKOHꢀ(50)/H
1%ꢀNaOHꢀ(50)/H
1%ꢀCsOHꢀ(50)/H
1%ꢀCa(OH)2ꢀ(50)/H
2
Oꢀ(50)ꢀ
Oꢀ(50)ꢀ
Oꢀ(50)ꢀ
Oꢀ(50)ꢀ
5ꢀ
5ꢀ
5ꢀ
7ꢀ
7ꢀ
8ꢀ
65.4ꢀ(48.9)dꢀ
63.8ꢀ
34.6ꢀ(28.8)d
36.2
2
2
66.1ꢀ
33.9
2
58.1ꢀ
41.9
1%ꢀKOHꢀ(50)/H
1%ꢀKOHꢀ(50)/H
2
Oꢀ(50)ꢀ
Oꢀ(50)ꢀ
60.3ꢀ
39.7
2
58.6ꢀ
41.4
a1u–wꢀ(10ꢀmmol);ꢀ addedꢀdropwiseꢀinꢀ1.0ꢀh;ꢀ GCꢀratio;ꢀ isolatedꢀyieldsꢀinꢀparenthesis.
c
d
easilyꢀ toꢀ giveꢀ cyclohexanolꢀ (2a)ꢀ inꢀ goodꢀ yieldꢀ underꢀ theꢀ
reactionꢀconditionsꢀ(Tableꢀ1,ꢀRunꢀ32).
Raneyꢀ Ni–Alꢀ alloyꢀ (5.0ꢀ g)ꢀ inꢀ waterꢀ (50ꢀ mL)ꢀ Afterꢀ beingꢀ heatedꢀ
forꢀ5ꢀhꢀatꢀ90ꢀ°C,ꢀtheꢀmixtureꢀwasꢀcooledꢀtoꢀroomꢀtemperatureꢀandꢀ
filtered through Celite. The residue was washed with ethyl acetate.ꢀ
The filtrate was neutralised with aq. hydrochloric acid, and the
resultingꢀmixtureꢀwasꢀextractedꢀwithꢀethylꢀacetate.ꢀTheꢀorganicꢀphaseꢀ
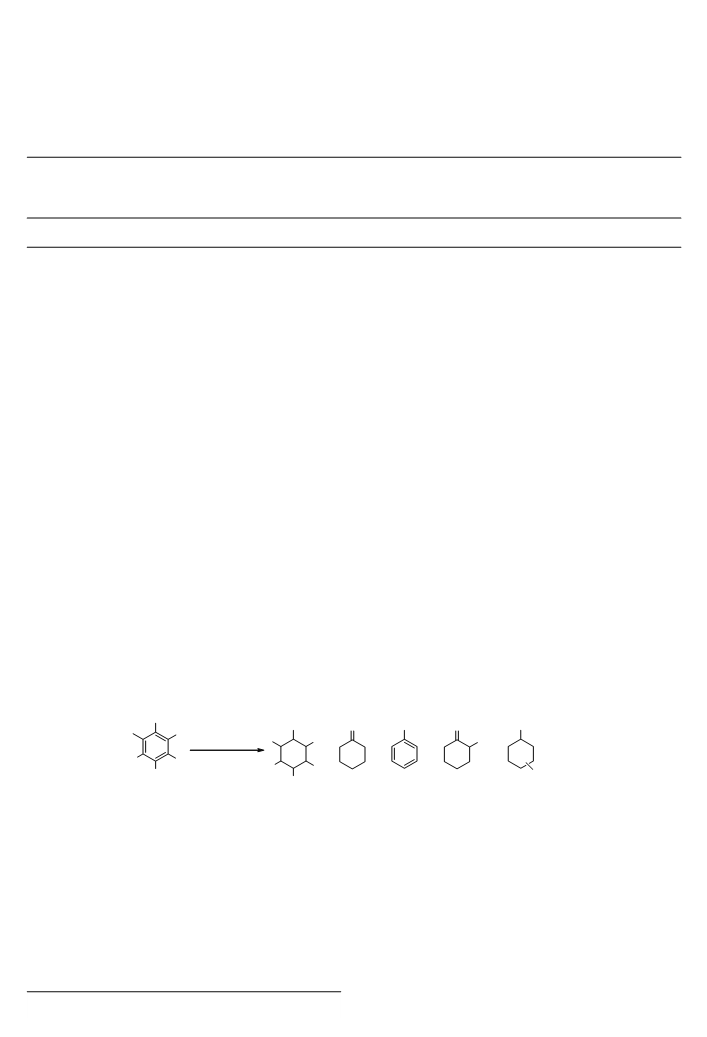
Hydrogenationꢀ ofꢀ trihydroxybenzenesꢀ withꢀ Raneyꢀ Ni–
Alꢀ alloyꢀ wasꢀ alsoꢀ studiedꢀ (Schemeꢀ 2ꢀ andꢀ Tableꢀ 2).ꢀ 1,3,5-
Trihydroxybenzeneꢀ(1u)ꢀ(phloroglucinol),ꢀwhenꢀreactedꢀwithꢀ
RaneyꢀNi–Alꢀalloyꢀ(500ꢀmg/mmolꢀsubstrate)ꢀuponꢀdropwiseꢀ
additionꢀ ofꢀ aꢀ 1wt%ꢀ aq.ꢀ NaOH,ꢀ KOH,ꢀ CsOHꢀ orꢀ Ca(OH)2ꢀ
solution,ꢀdidꢀnotꢀgiveꢀcyclohexane-1,3,5-triol,ꢀbutꢀaꢀmixtureꢀ
ofꢀcyclohexa-1,3-dioneꢀ(7)ꢀandꢀresorcinolꢀ(1g)ꢀinꢀaꢀratioꢀofꢀ
wasꢀ driedꢀ overꢀ anhydrousꢀ MgSO .ꢀ Afterꢀ removalꢀ ofꢀ theꢀ solvent,ꢀ
4
aꢀ mixtureꢀ ofꢀ cyclohexanolꢀ (2a),ꢀ cyclohexanoneꢀ (3),ꢀ phenolꢀ (4),ꢀ
2
-hydroxyꢀcyclohexanoneꢀ(5),ꢀandꢀcyclohexan-1,2-diolꢀ(6)ꢀinꢀaꢀratioꢀ
ofꢀ8.0:ꢀ8.9:ꢀ25.1:ꢀ6.3:ꢀ51.7ꢀwasꢀobtainedꢀ(Tableꢀ1,ꢀRunꢀ1).
For final identification, the compounds were separated by column
chromatographyꢀ onꢀ silicaꢀ gel,ꢀ whenꢀ mixturesꢀ ofꢀ structuresꢀ wereꢀ
obtainedꢀfromꢀtheꢀreactions.ꢀAllꢀofꢀtheꢀcompounds,ꢀ2a,ꢀ3, 3, 4, 5ꢀand 6ꢀ
wereꢀcomparedꢀwithꢀauthenticꢀsamplesꢀandꢀtheirꢀstructuresꢀwereꢀalsoꢀ
6
5.4%ꢀtoꢀ34.8%ꢀ(GC).ꢀAgain,ꢀtheꢀreductionꢀprogressedꢀlessꢀ
readilyꢀwhenꢀaꢀsmallerꢀamountꢀofꢀRaneyꢀNi–Alꢀalloyꢀ(400ꢀmg/ꢀ
mmolꢀsubstrate)ꢀwasꢀused,ꢀandꢀ1uꢀcouldꢀstillꢀbeꢀdetectedꢀ(15%,ꢀ
GC)ꢀatꢀtheꢀendꢀofꢀtheꢀreaction,ꢀevenꢀafterꢀtheꢀreactionꢀmixtureꢀ
hadꢀbeenꢀheatedꢀatꢀ90ꢀ°Cꢀforꢀ12ꢀh.ꢀSimilarly,ꢀtheꢀtribromoꢀandꢀ
trichloroꢀderivativesꢀ1v andꢀ1wꢀcouldꢀalsoꢀbeꢀreducedꢀuponꢀ
treatmentꢀwithꢀRaneyꢀNi–Alꢀalloyꢀinꢀanꢀaq.ꢀKOHꢀsolutionꢀtoꢀ
produceꢀaꢀmixtureꢀofꢀ7ꢀandꢀ1g.
In conclusion, we have developed a new and efficient
methodꢀforꢀtheꢀreductionꢀofꢀbromo-ꢀandꢀchlorophenolsꢀasꢀwellꢀ
asꢀofꢀdihydroxybenzenesꢀandꢀhalogenatedꢀdihydroxybenzenesꢀ
usingꢀcommerciallyꢀavailableꢀRaneyꢀNi–Alꢀalloyꢀinꢀaꢀdiluteꢀ
alkalineꢀ aqueousꢀ solution.ꢀ Noꢀ organicꢀ solventsꢀ wereꢀ usedꢀ
inꢀ theꢀ reactions.ꢀ Catecholsꢀ wereꢀ easilyꢀ reducedꢀ toꢀ affordꢀ aꢀ
mixtureꢀ ofꢀ cyclohexanolꢀ (2a),ꢀ cyclohexanoneꢀ (3),ꢀ phenolꢀ
1
assignedꢀonꢀtheꢀbasisꢀofꢀ HꢀNMR,ꢀIRꢀandꢀGC-MSꢀspectroscopicꢀdata.
1
8
2
max
-Methylcyclohexanolꢀ (2i, a mixture ofꢀ synꢀ and anti-isomers): ꢀ
-1
n
ꢀ(neat/cm )ꢀ3365ꢀ(bs,ꢀOH),ꢀ2930,ꢀ2870,ꢀ1450,ꢀ1062,ꢀ1045,ꢀ1030,ꢀ
9
78,ꢀ917,ꢀ840;ꢀd ꢀ(400ꢀMHz,ꢀCDCl )ꢀ1.02–1.88ꢀ(12H,ꢀm),ꢀ2.04ꢀ(1H,ꢀ
H 3
s,ꢀOH),ꢀ3.14ꢀ(0.81ꢀH,ꢀdt,ꢀJ =ꢀ4.0ꢀandꢀ9.2ꢀHz,ꢀCHOH, anti-isomer),ꢀ
3
.84ꢀ(0.19ꢀH,ꢀquintet,ꢀJ =ꢀ2.6ꢀHz,ꢀCHOH, syn-isomer);ꢀd ꢀ(67.8ꢀMHz,ꢀ
C
CDCl )ꢀforꢀsyn-isomer:ꢀ20.6,ꢀ24.5,ꢀ28.8,ꢀ30.9,ꢀ32.5,ꢀ35.8,ꢀ71.1;ꢀforꢀ
3
anti-isomer:ꢀ18.5,ꢀ25.2,ꢀ25.7,ꢀ33.6,ꢀ35.5,ꢀ40.2,ꢀ76.5;ꢀMSꢀ(EI,ꢀ70ꢀeV)ꢀ
+
m/zꢀ(%)ꢀ114ꢀ(M )ꢀ(38),ꢀ96ꢀ(100),ꢀ81ꢀ(88),ꢀ68ꢀ(64),ꢀ58ꢀ(58).
18
3
-Methylcyclohexanolꢀ(2j, a mixture of syn and anti-isomers): ꢀnmaxꢀ
-1
(
neat/cm )ꢀ3380ꢀ(bs,ꢀOH),ꢀ2940,ꢀ2880,ꢀ1452,ꢀ1105,ꢀ1045,ꢀ1030,ꢀ1000,ꢀ
9
45,ꢀ935;ꢀd ꢀ(400ꢀMHz,ꢀCDCl )ꢀ0.94–1.80ꢀ(12H,ꢀm),ꢀ2.02ꢀ(1H,ꢀs,ꢀOH),ꢀ
H 3
3.25ꢀ(0.80H,ꢀm,ꢀCHOH, anti-isomer),ꢀ3.76ꢀ(0.20H,ꢀm,ꢀCHOH, syn-
isomer);ꢀdCꢀ(67.8ꢀMHz,ꢀCDCl3)ꢀforꢀsyn-isomer:ꢀ20.2,ꢀ22.2,ꢀ26.6,ꢀ33.2,ꢀ
3
4.4,ꢀ41.6,ꢀ66.6;ꢀforꢀanti-isomer:ꢀ22.5,ꢀ24.4,ꢀ31.6,ꢀ34.3,ꢀ35.6,ꢀ44.9,ꢀ70.6;ꢀ
+
(
(
4),ꢀ 2-hydroxycyclohexanoneꢀ (5),ꢀ andꢀ cyclohexan-1,2-diolꢀ
6).ꢀResorcinolsꢀcouldꢀbeꢀconvertedꢀtoꢀcyclohexanolꢀinꢀhighꢀ
4
-1
(
neat/cm )ꢀ3370ꢀ(bs,ꢀOH),ꢀ2920,ꢀ1450,ꢀ1358,ꢀ1185,ꢀ1047,ꢀ980;ꢀd ꢀ(400ꢀ
H
yield.ꢀHydroquinonesꢀcouldꢀbeꢀtransformedꢀtoꢀcyclohexane-
,4-diolꢀinꢀgoodꢀyield.ꢀTheꢀadvantagesꢀofꢀtheꢀprocessꢀlieꢀinꢀtheꢀ
MHz,ꢀCDCl ) 0.86–1.78ꢀ(12H,ꢀm),ꢀ2.02ꢀ(1H,ꢀs,ꢀOH),ꢀ3.42ꢀ(0.78H,ꢀm,ꢀ
3
1
CHOH, anti-isomer),ꢀ 3.84–3.88ꢀ (0.22H,ꢀ m,ꢀ CHOH,ꢀ syn-isomer);ꢀ d ꢀ
C
(
6
7
easeꢀofꢀmanipulation,ꢀtheꢀshortꢀreactionꢀtimeꢀnecessary,ꢀandꢀ
theꢀmildnessꢀofꢀtheꢀreactionꢀconditions.ꢀRaneyꢀNi–Alꢀalloyꢀisꢀ
commerciallyꢀreadilyꢀavailableꢀandꢀis,ꢀofꢀcourse,ꢀcheaperꢀthanꢀ
theꢀRaneyꢀNiꢀcatalystꢀmadeꢀfromꢀit.ꢀFromꢀanꢀenvironmentalꢀ
stand-point,ꢀchlorinatedꢀandꢀbrominatedꢀphenolsꢀandꢀmixturesꢀ
thereofꢀ withꢀ theꢀ correspondingꢀ parentꢀ compoundsꢀ areꢀ
convertedꢀtoꢀtheꢀlessꢀtoxicꢀandꢀoftenꢀusefulꢀcyclohexanols.
3
Received 16 February 2009; accepted 19 March 2009
Paper 09/0450 doi: 10.3184/030823409X465394
Published online: 22 June 2009
References
ꢀ
Carbon,ꢀ2004,ꢀ42,ꢀ1371.
Experimental
General
ꢀ
IRꢀspectraꢀwereꢀmeasuredꢀwithꢀNicoletꢀFT-IRꢀ360,ꢀJASCOꢀIR-700ꢀ
andꢀNipponꢀDenshiꢀJIR-AQ2OMꢀmachines.ꢀ Hꢀandꢀ CꢀNMRꢀspectraꢀ
1
13
ꢀ
ꢀ
JP 2001300299.ꢀChem. Abstr.,ꢀ2001,ꢀ135,ꢀ334805.
6ꢀ D.ꢀSchmal,ꢀJ.ꢀvanꢀEnkelꢀandꢀP.J.ꢀvanꢀDuin,ꢀInst. Chem. Eng. Sympos. Ser.,ꢀ
1986,ꢀ98,ꢀ281.
1
wereꢀrecordedꢀwithꢀaꢀJEOLꢀEX-270ꢀspectrometerꢀ( Hꢀatꢀ270ꢀMHzꢀandꢀ
1
3
Cꢀatꢀ67.8ꢀMHz).ꢀTheꢀchemicalꢀshiftsꢀareꢀrelativeꢀtoꢀTMSꢀ(solventꢀ
CDCl ,ꢀunlessꢀotherwiseꢀnoted).ꢀMassꢀspectraꢀwereꢀmeasuredꢀwithꢀ
3
aꢀJMS-01-SG-2ꢀspectrometerꢀ[electronꢀimpactꢀmodeꢀ(EI),ꢀ70ꢀeVꢀorꢀ
fastꢀ atomꢀ bombardmentꢀ (FAB)]ꢀ andꢀ withꢀ aꢀ GC-MSꢀ 6890[GC]/HPꢀ
MS5973ꢀcombination.ꢀAnꢀHP-5ꢀGCꢀcolumnꢀfromꢀAgilentꢀwasꢀusedꢀ
ꢀ 7ꢀ P.P.ꢀCellier,ꢀJ.F.ꢀSpindler,ꢀM.ꢀTailleferꢀandꢀH.ꢀCristau,ꢀTetrahedron Lett.,ꢀ
2003,ꢀ44,ꢀ7191.
ꢀ
8ꢀ M.V.ꢀ Pera-Titus,ꢀ V.ꢀ Garcia-Mollina,ꢀ M.A.ꢀ Banos,ꢀ J.ꢀ Gimenezꢀ andꢀ
S.ꢀEsplugas,ꢀAppl. Catal. B: Environ.,ꢀ2004,ꢀ47,ꢀ219.
[50ꢀmꢀ¥ꢀ0.25ꢀmmꢀ¥ꢀ0.25ꢀmm;ꢀinjectorꢀtemperature:ꢀ300ꢀ°C;ꢀdetectorꢀ
ꢀ
temperature:ꢀ280ꢀ°C;ꢀstartingꢀtemperatureꢀ80ꢀ°C,ꢀholdingꢀtimeꢀ3ꢀmin,ꢀ
temperatureꢀrisingꢀrate:ꢀ10ꢀ°C/min; final temperature 300ꢀ°C;ꢀholdingꢀ
timeꢀ10ꢀmin,ꢀcarrierꢀgas:ꢀhelium].
ꢀ11ꢀ G.ꢀYuanꢀandꢀM.A.ꢀKean,ꢀChem. Eng. Sci.,ꢀ2003,ꢀ58,ꢀ257.
ꢀ12ꢀ E.-J.ꢀShinꢀandꢀM.A.ꢀKeane,ꢀJ. Hazard. Mater., 1999,ꢀ66,ꢀ265.
TheꢀRaney-Niꢀalloyꢀ[Ni/Alꢀ50:50wt%]ꢀwasꢀacquiredꢀcommerciallyꢀ
fromꢀ Jinzhouꢀ Catalystꢀ Companyꢀ (16,ꢀ Wenshengli,ꢀ Linhe,ꢀ Jinzhouꢀ
1
2100,ꢀ P.R.ꢀ China).Theꢀ halogenatedꢀ phenolsꢀ wereꢀ purchasedꢀ fromꢀ
Sino-PharmaꢀChemicalꢀReagentꢀCo.,ꢀLtd.,ꢀShanghaiꢀ200001,ꢀChina.
ꢀ
16ꢀ P.ꢀ Makowski,ꢀ R.ꢀ Demir-Cakan,ꢀ M.ꢀ Antonietti,ꢀ F.ꢀ Goettmannꢀ andꢀ
Reduction of chlorophenols
Aꢀ1wt%ꢀaq.ꢀKOHꢀsolutionꢀ(100ꢀmL)ꢀwithinꢀ1.0ꢀhꢀandꢀatꢀ90ꢀ°Cꢀwasꢀ
addedꢀ dropwise.ꢀ Toꢀ aꢀ suspensionꢀ ofꢀ 1aꢀ (10ꢀ mmol,ꢀ 1.10ꢀ g),ꢀ andꢀ
M.-M.ꢀTitirici,ꢀJ. Chem. Soc.,ꢀChem. Commun.,ꢀ2008,ꢀ999.
ꢀ17ꢀ G.-B.ꢀLiu,ꢀM.ꢀTashiroꢀandꢀT.ꢀThiemann, Tetrahedron, 2009, 65,ꢀ2497.
ꢀ18ꢀ S.L.ꢀTan,ꢀG.-B.ꢀLiu,ꢀX.ꢀGaoꢀandꢀT.ꢀThiemann,ꢀJ. Chem. Res.,ꢀ2009,ꢀ5.

 Liu, Guo-Bin
Liu, Guo-Bin
 Zhao, Hong-Yun
Zhao, Hong-Yun
 Zhang, Jie
Zhang, Jie
 Thiemannb, Thies
Thiemannb, Thies