868
N. Kongkathip, B. Kongkathip, and N. Noimai
(d, J ¼ 7.0 Hz, 3H, C-21), 1.05 (s, 3H, C-18), 2.03 (s, 3H, AcO), 2.41
(t, J ¼ 8.0 Hz, 1H, C-9), 2.45 (m, 1H, C-9), 3.45–3.33 (m, 2H, C-26),
4.40–4.35(m, 1H, C-16), 4.65 (m, 1H, C-3). MS (FAB) m/z (relative
intensity) 473 (Mþ þ 1, 100), 431 (3.57), 413 (9.70), 395 (9.70).
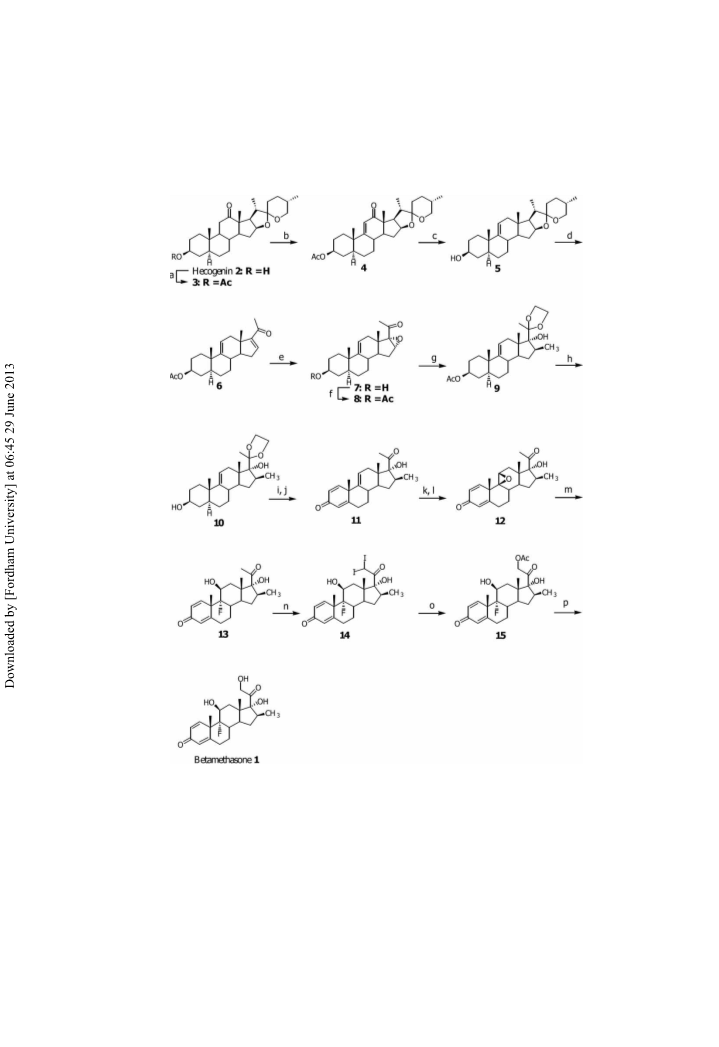
D9(11)22-Isoallospirosten-3b-ol-12-one-3-acetate (4): A mixture of m-iodoxy-
benzoic acid (45 g, 160.71mmol) and diphenyl diselenide (4.95g,
15.76 mmol) was heated under reflux in toluene (500mL) until the yellow
color of the diselenide disappeared. To the mixture, a solution of 3 (25 g,
52.85 mmol) in toluene (50 mL) was added. The reaction mixture was further
heated under reflux for 12 h and cooled to room temperature. The reaction
mixture was washed with water and saturated sodium hydrogen carbonate
solution, and then the organic layer was dried over anhydrous sodium
sulphate. The organic solvent was evaporated under vacuum and then recrystal-
lized from ethyl acetate, which afforded 4 as a white crystal (17.92g, 72%), mp
218–220 8C. The aqueous phase was acidified with diluted sulfuric acid to give
the precipitate of m-iodobenzoic acid. FTIR (KBr) nmax 1734, 1671, 1596cm21
.
1H NMR (CDCl3, 400 MHz) d: 0.8 (s, 3H, C-19), 0.92 (d, J ¼ 6.0 Hz, 3H, C-27),
1.1 (s, 3H, C-21), 1.11 (s, 3H, C-18), 2.05 (s, 3H, AcO), 3.35 (t, J ¼ 11.6Hz, 1H,
C-26), 3.50 (bd, J ¼ 11.2Hz, 1H, C-26), 4.40 (q, J ¼ 8 Hz, C-16), 4.65 (m, 1H,
C-3), 5.7 (s, 1H, C-11). 13C NMR (CDCl3, 100 MHz) d: 204.8, 170.5, 120.1,
109.4, 79.7, 72.6, 66.9, 53.7, 52.4, 51.0, 42.5, 42.4, 39.2, 36.8, 34.5, 33.8,
32.4, 31.5 (2C), 31.4, 30.2, 28.8, 27.5, 27.2, 21.3, 18.4, 17.1, 15.0, 13.1ppm.
MS (FAB) m/z (relative intensity) 470 (Mþ, 11.76), 442 (7.35), 411 (10.92),
398 (11.03), 356 (38.97), 332 (27.94), 304 (26.47), 259 (22.79), 248 (33.82),
159 (26.47), 139 (100), 121 (54.41), 69 (80.14), 43 (92.64), 29 (98.52).
D9(11)22-Isoallospirosten-3b-ol (5): A mixture of 4 (10 g, 21.28 mmol),
ethylene glycol (225 mL), and 85% hydrazine hydrate (75 mL) was refluxed
for 1 h, cooled, and then treated with potassium hydroxide (25 g, 625 mmol)
and 10 mL of water. The reaction mixture was heated to 195–197 8C and
refluxed for 5 h. After neutralization with 10% hydrochloric acid, the
resulting mixture was extracted with methylene chloride, washed with water,
and dried over anhydrous sodium sulphate. After removal of the solvent, 5
(8.37 g, 95%) was recrystallized from ethyl acetate as a white crystal, mp
216–217 8C. FTIR (KBr) nmax 3262, 1663 cm21
.
1H NMR (CDCl3,
400 MHz)d: 0.8 (s, 3H, C-19), 0.97 (s, 3H, C-27), 0.98 (s, 3H, C-21),
1.11(s, 3H, C-18), 3.4 (t, J ¼ 8 Hz, 1H, C-26), 3.50 (d, J ¼ 11.6 Hz, 1H,
C-26), 3.6 (m, 1H, C-3), 4.45 (q, J ¼ 6 Hz, 1H, C-16), 5.3 (bs, 1H, C-11).
13C NMR (CDCl3, 100 MHz) d: 147.1, 115.9, 109.3, 80.9, 71.0, 66.9, 61.4,
53.8, 43.1, 42.1, 41.8, 38.8, 38.3, 37.8, 35.7, 35.4, 33.1, 32.8, 31.5, 31.2,
30.3, 28.7, 28.4, 17.9, 17.1, 15.8, 14.1 ppm. MS (FAB) m/z (relative
intensity) 414 (Mþ, 3.9), 271 (8.5), 213 (9.0), 185 (85), 106 (26.0), 93 (100).
3b-Acetoxy-5a-pregn-9(11),16-diene-20-one (6): 1) A mixture of 5 (4.33 g,
10.45 mmol), acetic anhydride (40 mL), pyridine (1.648 mL), and ammonium

 Kongkathip, Ngampong
Kongkathip, Ngampong
 Kongkathip, Boonsong
Kongkathip, Boonsong
 Noimai, Naratitt
Noimai, Naratitt