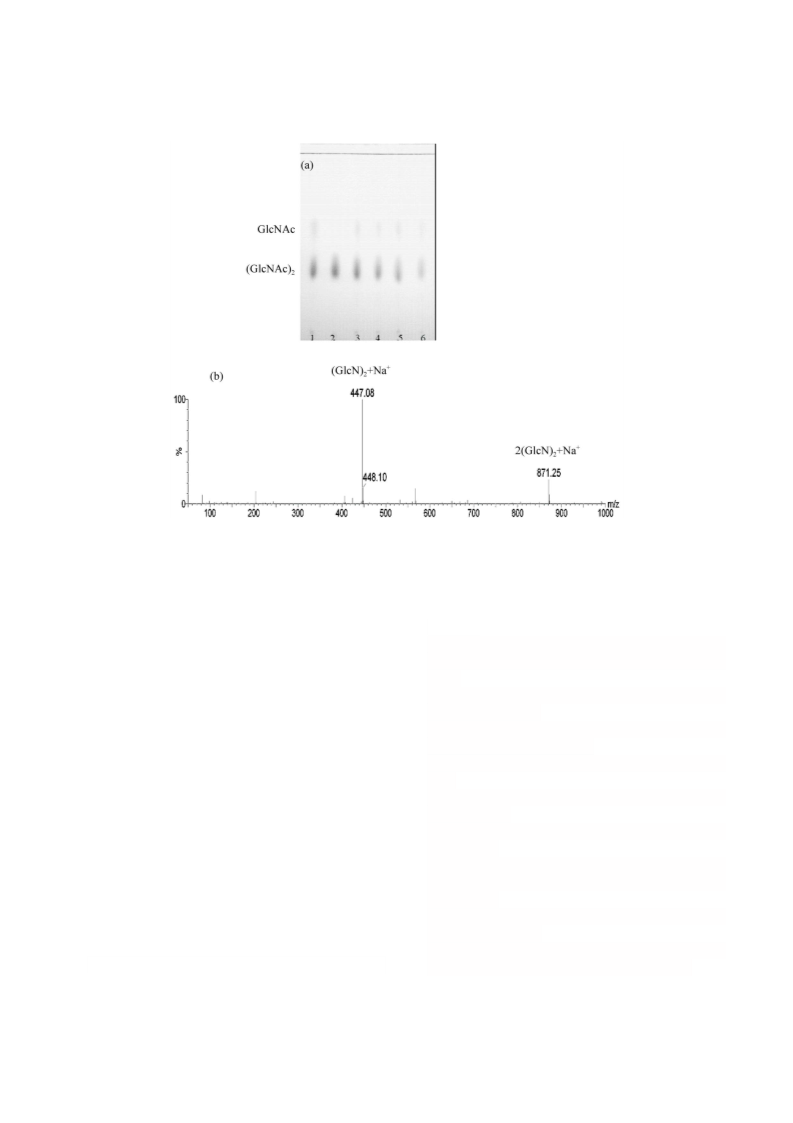
Large-scale Preparation of N,N-Diacetyl Chitobiose
J. Chin. Chem. Soc., Vol. 56, No. 4, 2009 689
yield. Although the chemical degradation of chitin is cost-
less, the laborious separation can only allow a small-scale
isolation of N,N-diacetyl chitobiose or other chito-oligo-
saccharides. Although many researchers have shown some
drawbacks, such as product inhibition, protracted reaction4
and the contamination of monomer, in an enzymatic pro-
cess, an easily obtained chitinase with a high product speci-
ficity was expected to be useful for the preparation of
N,N-diacetyl chitobiose. We report here that a chiA gene
from S. marcescens was cloned, over-expressed and ap-
plied to preparation of N,N-diacetyl chitobiose on a 100-g
scale.
min at 7000 xg. The cell pellet was resupended in sodium
phosphate buffer (10 mL, 20 mM, pH 7.0) and then sub-
jected to cell disruption with ultrasonication. After centrif-
ugation to remove cell debris, the supernatant (~10 mL)
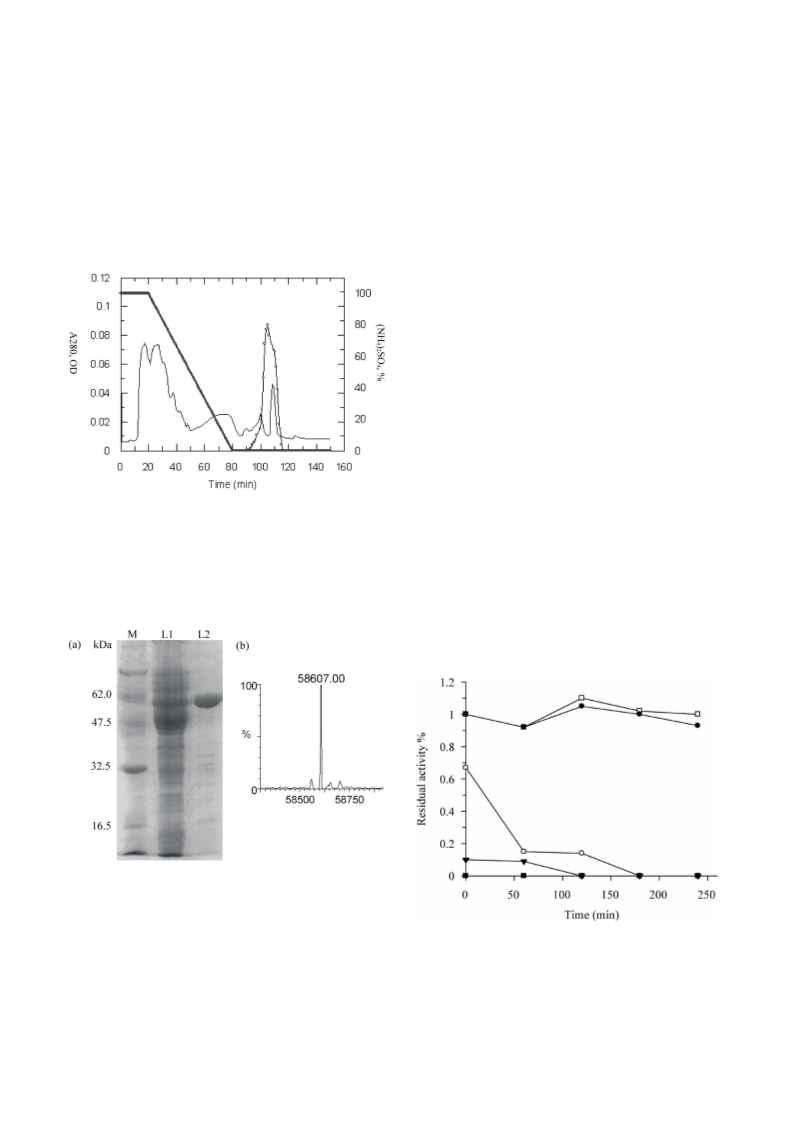
was loaded onto a hydrophobic interaction column (2.4 ´
20 cm, high-performance phenylsepharose), which was
pre-equilibrated with sodium phosphate buffer (20 mM,
pH 7.0) containing (NH4)2SO4 (1 M). Elution at flow rate 2
mL/min with a linear gradient of (NH4)2SO4 from 1000 to 0
mM was performed at a decreasing rate 16.67 mM/min.
Fractions with chitinase activity were collected and loaded
onto a series connection of a cation-exchange column (5
mL HiTrap) and an anion-exchange (5 mL HiTrap) column,
pre-equilibrated with phosphate buffer (20 mM, pH 8.0).
Chromatography was performed with isocratic elution and
phosphate buffer (20 mM, pH 8.0) at a flow rate 2.0 mL/
min. The active fractions were pooled for further study. All
purification steps were performed at ambient temperature
(25 °C).
EXPERIMENTAL SECTION
Material
Chemical and microbes. Buffers Sigma-Aldrich (St.
Louis, MO, USA), HiTrap SP and Q columns and phenyl-
sepharose resin (Amersham Bioscience, Uppsala, Swe-
den), chitin, (local supplier in Taiwan) and S. marcescens
(ATCC 990, Food Industry Research and Development In-
stitute, Hsinchu, Taiwan) were obtained from the indicated
sources.
Colloidal chitin preparation
Chitin powder (100 g) was suspended in concentrated
HCl (600 mL) and incubated at room temperature until
chitin powder was completely dissolved (about 3 h). The
resulting solution was then poured into doubly deionized
water (4 L) with rapid stirring to form the precipitant of col-
loidal chitin, which was subsequently collected by centrif-
ugation at 7000 xg for 10 min at 4 °C. The precipitant was
washed with sterile distilled water (200 mL each) several
times to bring the pH value of the colloidal chitin suspen-
sion to 2.0-3.0. After neutralization with sodium hydroxide
(1 M), the low-salt colloidal chitin was obtained on re-
peated centrifugation (7000 xg, 10 min) and washing with
water 2-3 times. The acid-treated chitin was kept at 4 °C as
a pellet for further applications.
Cloning of chiA
The chiA gene was PCR-amplified from genomic
DNA of S. marcescens using primers 5’-GGAATCAC-
ATATGCGCAAATTTAA-3’, and 5’-GCAACCGATTAT-
TGAACGCCGG-3’ which were designed on the basis of
the S. marcescens chiA gene published in the GenBank da-
tabase (accession number AF085718). PCR amplification
was performed with Vent DNA polymerase (NEB) with 25
cycles; each cycle involved 94 °C, 30 s for denaturation, 60
°C, 30 s for annealing, and 72 °C, 4 min for extension. The
PCR fragment was first cloned into a cloning vector
(QuanTox, Stratagen Co.) and sequenced. The correct gene
was then inserted into Nde I/EcoR I sites of pRSET A and
expressed in Escherichia coli.
Enzyme activity assay
Chitinase activity was measured via estimating the re-
ducing ends of sugars. The assay was performed by mixing
colloidal chitin (0.25 mL, 1%, pH 7.0), and suitably diluted
enzyme (0.25 mL) for 1 h at 37 °C. The catalytic reaction
was terminated and analyzed on adding dinitrosalicylic
acid reagent12 (0.5 mL). The mixture was boiled for 15 min,
chilled and centrifuged to remove insoluble chitin. The re-
sulting adduct of reducing sugars were measured spectro-
photometrically at 540 nm. One unit of chitinase activity is
defined as the amount of enzyme required to release detect-
Cultural conditions and purification of Chitinase A
(ChiA)
E. coli BL21 (DE3) served as the host strain for pro-
tein expression. A single colony was inoculated into LB
medium (5 mL) containing ampicillin (0.1 mg/mL) and cul-
tured at 37 °C on a rotary shaker for 12 h. The overnight
culture was then transferred into a conical flask (2 L) con-
taining LB medium (1 L) with ampicillin (0.1 mg/mL) at 37
°C for 15 h. The culture broth was centrifuged at 4 °C for 10

 Wu, Yue-Jin
Wu, Yue-Jin
 Cheng, Chih-Yu
Cheng, Chih-Yu
 Li, Yaw-Kuen
Li, Yaw-Kuen