
Chemical and Pharmaceutical Bulletin p. 843 - 846 (2018)
Update date:2022-08-16
Topics:
 Tsubogo, Tetsu
Tsubogo, Tetsu
 Aoyama, Saki
Aoyama, Saki
 Takeda, Rika
Takeda, Rika
 Uchiro, Hiromi
Uchiro, Hiromi
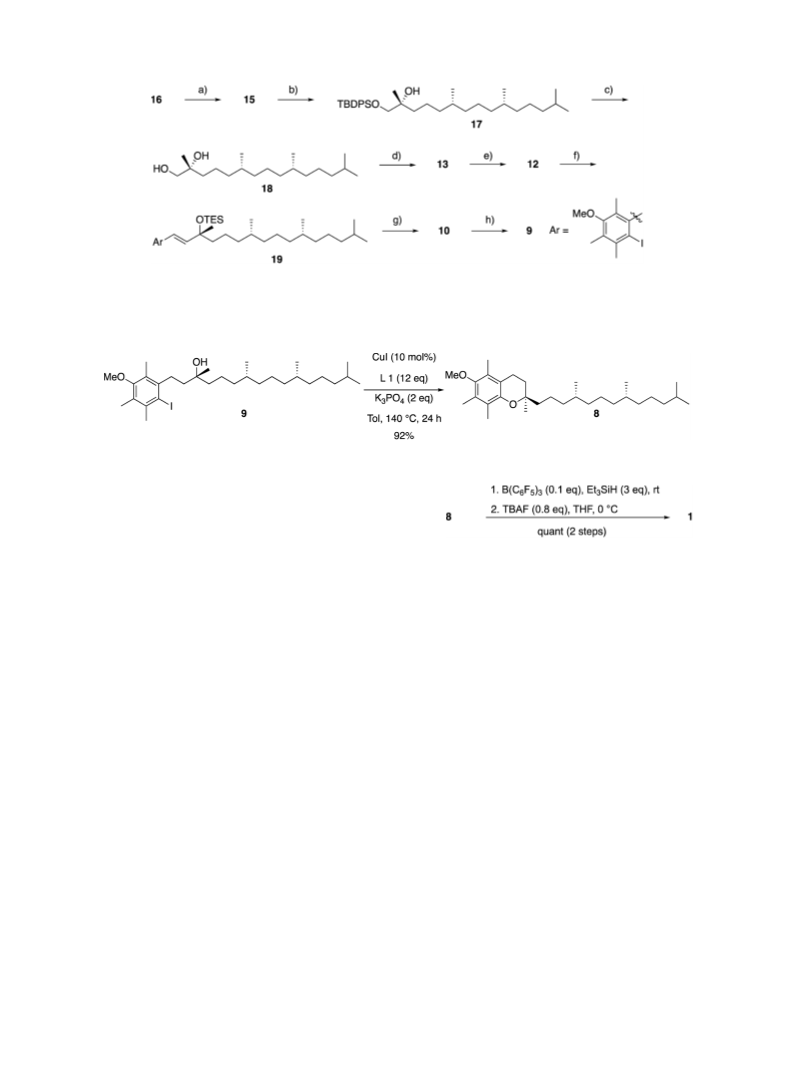
The complete synthesis of D-α-tocopherol was achieved using our developed-Ullmann C–O coupling reaction as a key reaction. The synthesis of the core structure of D-α-tocopherol, which is a chiral chromane, has never been reported using intramolecular Ullmann C–O coupling reactions owing to the low reactivity of electron-rich iodoarenes with tertiary alcohols. Because the developed intramolecular C–O coupling reactions prefer electron-rich iodoarenes with tertiary alcohols, we successfully synthesized the chiral chromane core and achieved the total synthesis of D-α-tocopherol.
View More

Changzhou Kingyo Chemical Corporation Ltd.
website:http://www.kingyochem.com
Contact:+86-519-85105717
Address:19# Wuqing North Road, Changzhou , Jiangsu, China
Tianjin Pharmacn Medical Technology Co.,Ltd.
Contact:86-22-60122566ext.866(English),23359620
Address:Green Industrial Base, 6 Haitaifazhan Sixth Rd., Huayuan Industrial Area, Tianjin, 300384, China
Shanghai Sunwise Chemical Co., Ltd
website:http://www.sunwisechem.com
Contact:86-021-33883180
Address:Room 10E, Building G, Westlink Center, No. 2337 Gudai Road, Minhang District, Shanghai, China PC: 201100
Tangshan Wisdom Trading Co.,Ltd
Contact:86 315 2222979
Address:No.41 BeiXinXi Road, Yangguang building 1-1102 , Tangshan, Hebei, China
Chengdu Sino-Strong Pharmaceutical Co.,Ltd.
website:http://www.sino-strong.com.cn
Contact:+86-28-82666753
Address:459 West haike road,Cross-straits technological industry park, Wenjiang district,Chengdu, P.R.China
Doi:10.1002/pola.24258
(2010)Doi:10.1002/jms.1190300406
(1995)Doi:10.1016/j.phytochem.2012.07.006
(2012)Doi:10.1002/adsc.200404197
(2005)Doi:10.1002/chem.201402798
(2014)Doi:10.1016/j.jorganchem.2013.06.037
(2013)