Dalton Transactions
Paper
III
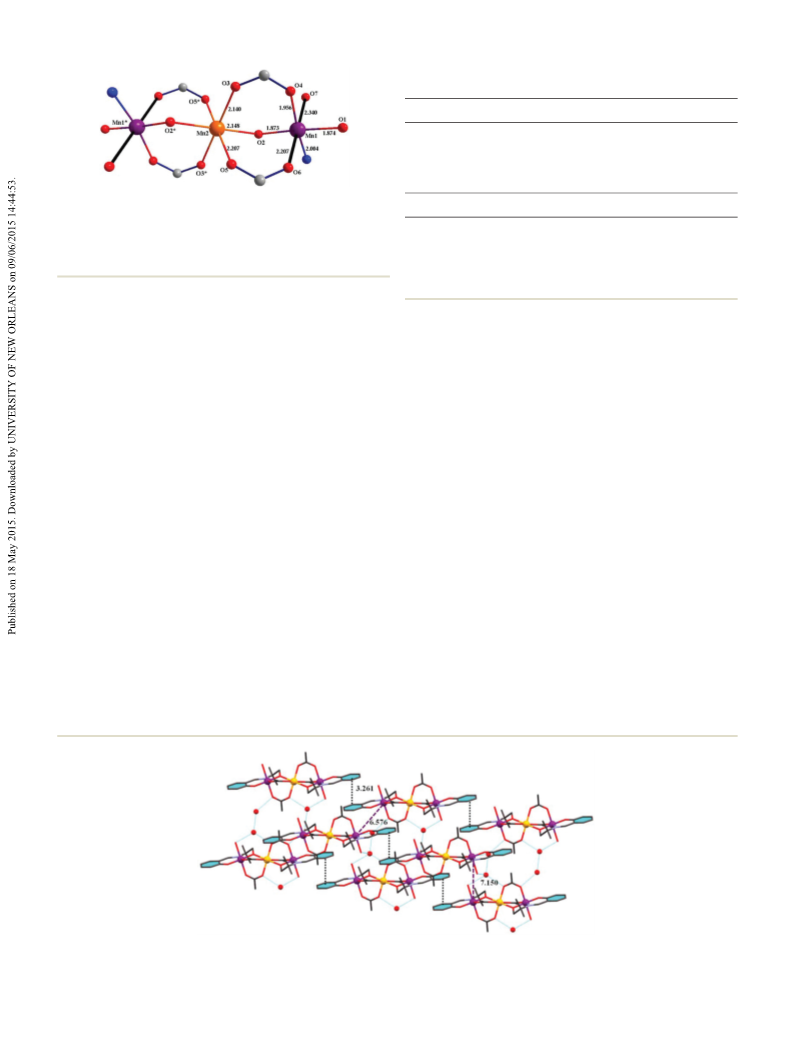
peripheral Mn ions, which leads to the S = 3/2 ground state.
5988–5998; (d) V. K. Yachandra, K. Sauer and M. P. Klein,
Chem. Rev., 1996, 96, 2927–2950.
5 V. L. Pecoraro, Manganese Redox Enzymes, VCH, New York,
1992.
6 A. Zouni, H. T. Witt, J. Kern, P. Fromme, N. Krauss,
W. Saenger and P. Orth, Nature, 2001, 409, 739–743.
7 N. Kamiya and J. R. Shen, Proc. Natl. Acad. Sci. U. S. A.,
2003, 100, 98–103.
8 (a) K. N. Ferreira, T. M. Iverson, K. Maghlaoui, J. Barber
and S. Iwata, Science, 2004, 303, 1831–1838; (b) J. Biesiadka,
B. Loll, J. Kern, K.-D. Irrgang and A. Zouni, Phys. Chem.
Chem. Phys., 2004, 6, 4733–4736.
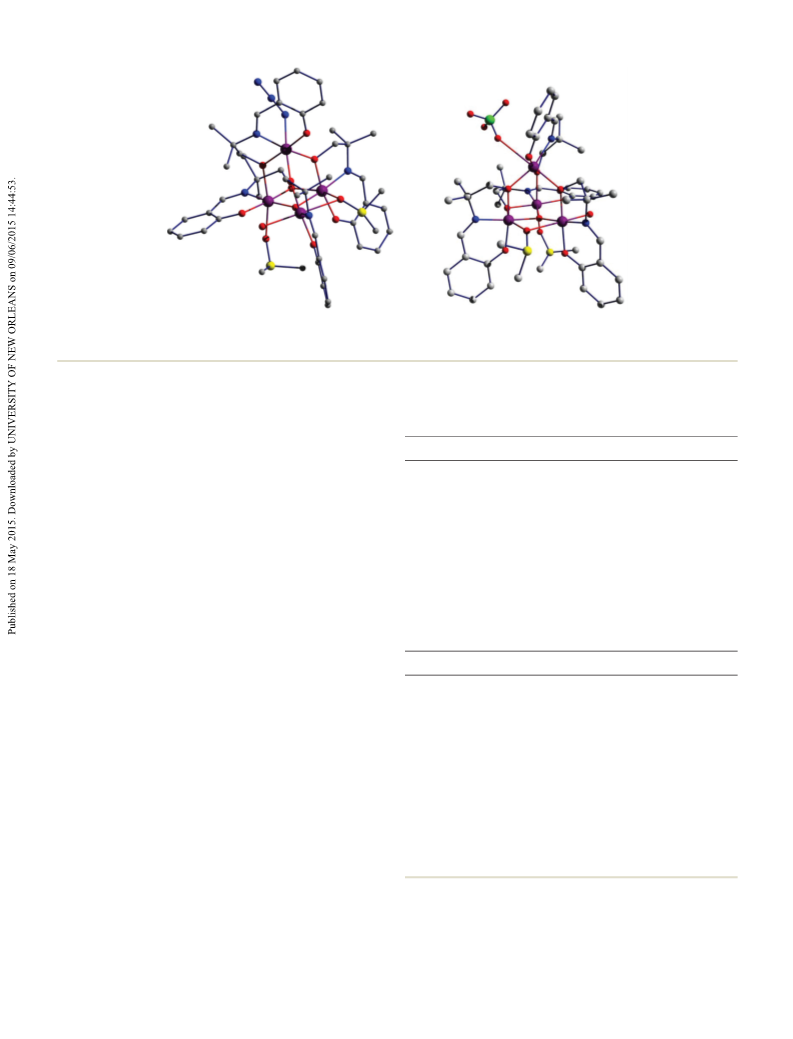
A comparison to the related trimers found in the literature
revealed that the strength of this exchange coupling correlates
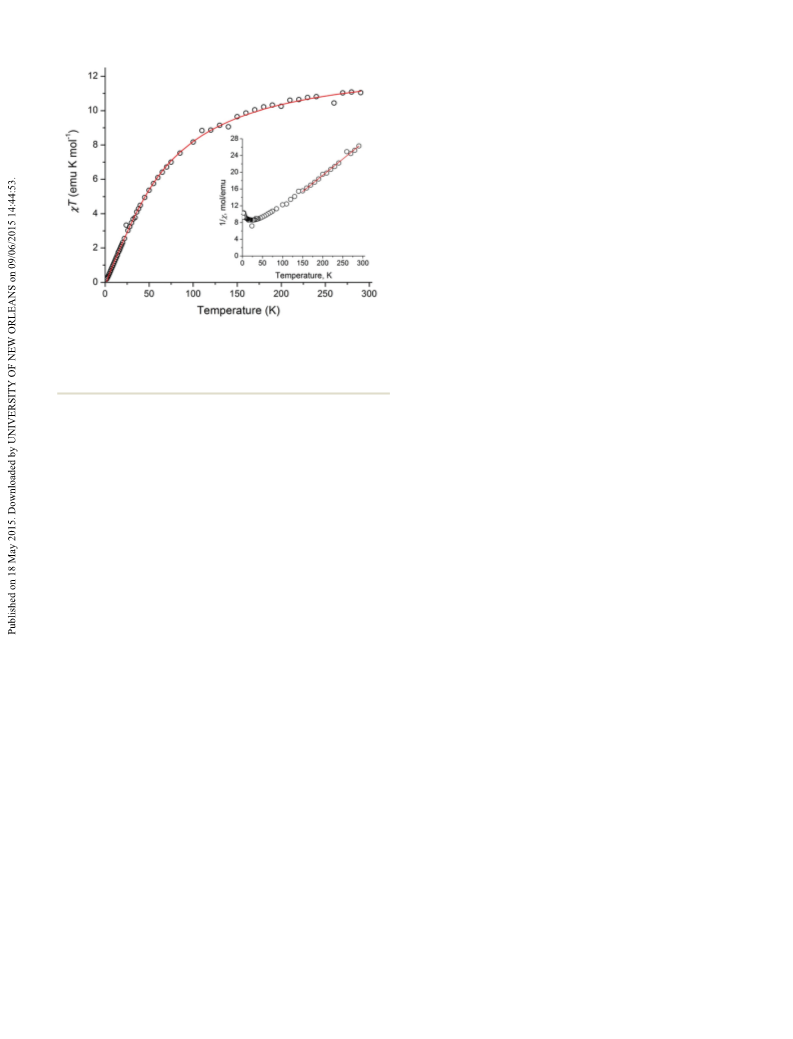
well with the Mn ⋯Mn separation. The magnetic behavior
of the tetranuclear complex 3 is primarily dependent on the
orientation of the elongated Jahn–Teller axes of the four octa-
II
III
III
hedrally coordinated Mn ions. As a result, magnetism is
dominated by antiferromagnetic exchange in two different
pairs of ions, leading to the overall singlet ground state.
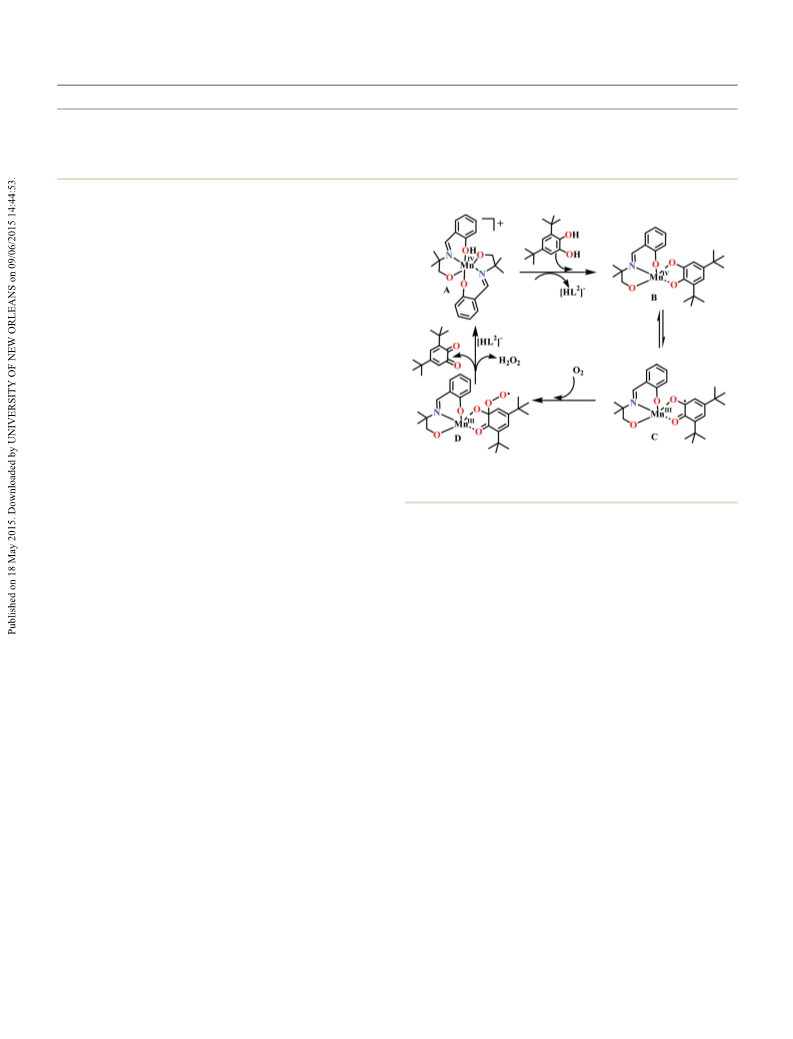
Kinetic studies in solution revealed that both the trinuclear
and tetranuclear complexes are efficient catalysts for the
2
solvent dependent oxidation of 3,5-di-tert-butylcatechol by O ,
with complex 2 being the most active. In MeOH medium, all
complexes are characteristically inactive. In MeCN, the reactiv-
9 S.-Y. Chen, C. C. Beedle, P.-R. Gan, G.-H. Lee, S. Hill and
E.-C. Yang, Inorg. Chem., 2012, 51, 4448–4457.
ity for the oxidation of 3,5-DTBC increases in the order 4 < 3 < 10 A. Saha, K. A. Abboud and G. Christou, Inorg. Chem., 2011,
< 2. The ESI-MS (positive) evidences suggest the presence of 50, 12774–12784.
metal complex–catechol substrate aggregates with bidentate 11 P.-P. Yang, C.-Y. Shao, L.-L. Zhu and Y. Xu, Eur. J. Inorg.
1
catechol units to Mn centers.
Chem., 2013, 5288–5296.
12 L. Zhang, C. I. Onet, R. Clérac, M. Rouzières, B. Marzec,
M. Boese, M. Venkatesan and W. Schmitt, Chem. Commun.,
2
013, 49, 7400–7402.
Acknowledgements
1
3 P. Kar, Y. Ida, T. Kanetomo, M. G. B. Drew, T. Ishida and
We are very much thankful to the reviewers at the revision
A. Ghosh, Dalton Trans., 2015, 44, 9795–9804.
stage for their valuable and most appropriate suggestions in 14 (a) P. Wang, G. P. A. Yap and C. G. Riordan, Chem.
improving the standard of the manuscript. MP is thankful to
the Council of Scientific and Industrial Research, New Delhi,
India, for financial support toward her doctoral degree. The
authors give thanks to DST, New Delhi, for providing the
Commun., 2014, 50, 5871–5873; (b) S. K. Dey and
A. Mukherjee, New J. Chem., 2014, 38, 4985–4995;
(c) A. Biswas, L. K. Das, M. G. B. Drew, G. Aromí, P. Gamez
and A. Ghosh, Inorg. Chem., 2012, 51, 7993–8001.
Single Crystal X-ray Diffractometer facility in the Department 15 G. Maayan and G. Christou, Inorg. Chem., 2011, 50, 7015–
of Chemistry, IIT Kharagpur under its FIST program. MP also 7021.
gives thanks to Dr Debiranjan Tripathy for fruitful discussion. 16 (a) M. Dey, C. P. Rao, P. K. Saarenketo and K. Rissanen,
The study on the molecular magnetism by the FSU group was
partially supported by the National Science Foundation (award
NSF-0911109).
Inorg. Chem. Commun., 2002, 5, 380–383; (b) M. Dey,
C. P. Rao, P. K. Saarenketo, K. Rissanen, E. Kolehmainen
and P. Guionneau, Polyhedron, 2003, 22, 3515–3521;
(
c) M. Dey, C. P. Rao, P. K. Saarenketo, K. Rissanen and
E. Kolehmainen, Eur. J. Inorg. Chem., 2002, 2207–2215;
d) M. Dey, C. P. Rao, P. K. Saarenketo and K. Rissanen,
Inorg. Chem. Commun., 2002, 5, 924–928.
(
References
1
G. E. Kostakis, A. M. Ako and A. K. Powell, Chem. Soc. Rev., 17 G. A. Bain and J. F. Berry, J. Chem. Educ., 2008, 85, 532–535.
2
010, 39, 2238–2271.
18 Saint, Smart and XPREP, Siemens Analytical X-ray Instru-
2
(a) A. M. Mowson, T. N. Nguyen, A. K. Abboud and
ments Inc., Madison, WI, 1995.
G. Christou, Inorg. Chem., 2013, 52, 12320–12322; 19 G. M. Sheldrick, SADABS Software for Empirical Absorption
(
2
b) M. Sarkar, V. Bertolasi and D. Ray, Eur. J. Inorg. Chem.,
010, 2530–2536.
(a) M. T. Caudle, J. W. Kamp, M. L. Kirk, P. G. Rasmussen
Correction, University of Göttingen, Institute fur Anorga-
nische Chemieder Universitat, Göttingen, Germany,
1999–2003.
3
4
and V. L. Pecoraro, J. Am. Chem. Soc., 1997, 119, 9297–9298; 20 (a) G. M. Sheldrick, SHELXS-97, University of Göttingen,
(
b) F. Habib, G. Brunet, F. Loiseau, T. Pathmalingam,
Göttingen, Germany, 1997; (b) G. M. Sheldrick, SHELXL 97,
Program for Crystal Structure Refinement, University of
Göttingen, Göttingen, Germany, 1997.
T. J. Burchell, A. M. Beauchemin, W. Wernsdorfer,
R. Clérac and M. Murugesu, Inorg. Chem., 2013, 52, 1296–
1
303.
21 (a) H. Miyasaka, K. Nakata, L. Lecren, C. Coulon,
Y. Nakazawa, T. Fujisaki, K. Sugiura, M. Yamashita and
R. Clérac, J. Am. Chem. Soc., 2006, 128, 3770–3783;
(b) A. Das, K. Gieb, Y. Krupskaya, S. Demeshko, S. Dechert,
R. Klingeler, V. Kataev, B. Büchner, P. Müller and F. Meyer,
J. Am. Chem. Soc., 2011, 133, 3433–3443; (c) M. Hirotsu,
Y. Shimizu, N. Kuwamura, R. Tanaka, I. Kinoshita,
(a) K. N. Ferreira, T. M. Iverson, K. Maghlaoui, J. Barber
and S. Iwata, Science, 2004, 303, 1831–1838;
(
b) T. G. Carrell, A. M. Tyryshkin and G. C. J. Dismukes,
Biol. Inorg. Chem., 2002, 7, 2–22; (c) R. M. Cinco,
A. Rompel, H. Visser, G. Aromí, G. Christou, K. Sauer,
M. P. Klein and V. K. Yachandra, Inorg. Chem., 1999, 38,
This journal is © The Royal Society of Chemistry 2015
Dalton Trans.

 Pait, Moumita
Pait, Moumita
 Shatruk, Michael
Shatruk, Michael
 Ray, Debashis
Ray, Debashis