Please do not adjust margins
Organic & Biomolecular Chemistry
Page 4 of 4
COMMUNICATION
Journal Name
providing the correctly folded somatostain with an isolated
yield of 68% (Fig. 4).
2007, 2, 457-468.
2
3
(a) K. Holland-Nell and M. Meldal, AngeDwO.IC: h10e.m10.3I9n/tC.9EOdB.,021101141E,
50, 5204-5206; (b) K. Hu, H. Geng, Q. Zhang, Q. Liu, M. Xie, C.
Sun, W. Li, H. Lin, F. Jiang, T. Wang, Y.-D. Wu and Z. Li, Angew.
Chem. Int. Ed., 2016, 55, 8013-8017; (c) T. Wang, J. Fan, X.-X.
Chen, R. Zhao, Y. Xu, D. Bierer, L. Liu, Y.-M. Li, J. Shi and G.-M.
Fang, Org. Lett., 2018, 20, 6074–6078; (d) D.-L. Huang, J.-S. Bai,
M. Wu, X. Wang, B. Riedl, E. Pook, C. Alt, M. Erny, Y.-M. Li, D.
Bierer, J. Shi and G.-M. Fang, Chem. Commun., 2019, 55, 2821-
2824.
(a) Y. Fujiwara, K. Akaji and Y. Kiso, Chem. Pharm. Bull., 1994,
42, 724-726; (b) J. Hansen, F. Diness and M. Meldal, Org.
Biomol. Chem., 2016, 14, 3238-3245; (c) H. E. Elashal, Y. E. Sim
and M. Rai, Chem. Sci., 2017, 8, 117-123; (d) H. E. Elashel, R.
D. Cohen and M. Raj, Chem. Commun., 2016, 52, 9699-9702;
(e) Y. Han, F. Albericio and G. Barany, J. Org. Chem., 1997, 62,
4307-4312.
(a) J. Lukszo, D. Patterson, F. Albericio and S. A. Kates, Lett.
Pept. Sci., 1996, 3, 157−166; (b) S. Ni, H. Zhang, W. Huang, J.
Zhou, H. Qian and W. Chen, J. Pept. Sci., 2010, 16, 309-313.
(a) Z. Huang, D. J. Derksen and J. C. Vederas, Org. Lett., 2010,
12, 2282-228; (b) V. Diaz-Rodríguez, D. G. Mullen, E. Ganusove,
J. M. Becker and M. D. Distefano, Org. Lett., 2012, 14, 5648-
5651; (c) I. Ramos-Tomillero, H. Rodríguez and F. Albericio,
Org. Lett., 2015, 17, 1680-1683; (d) V. Juvekar and Y. D. Gong,
Org. Lett., 2016, 18, 836-839; (e) D. Lelièvre, V. P. Terrier, A.
F. Delmas and V. Aucagne, Org. Lett., 2016, 18, 920-923.
(a) G.-M. Fang, Y.-M. Li, F. Shen, Y.-C. Huang, J.-B. Li, Y. Lin, H.-
K. Cui and L. Liu, Angew. Chem. Int. Ed., 2011, 50, 7645-7649;
(b) G.-M. Fang, J.-X. Wang and L. Liu, Angew. Chem. Int. Ed.,
2012, 51, 10347-10350; (c) D. T. Flood, J. C. J. Hintzen, M. J.
Bird, P. A. Cistrone, J. S. Chen and P. E. Dawson, Angew. Chem.
Int. Ed., 2018, 57, 11634-11639.
Furthermore, by using the same strategy, we also obtained
the host-defense skin peptide Riparin 1.1b containing one
disulfide bond (Fig. S12).13 As expected, chemical synthesis of
Riparin 1.1b was smooth. Riparin 1.1b hydrazide was readily
prepared from the hydrazine-trityl(2-Cl) resin in an isolated
yield of 73%. Riparin 1.1b hydrazide was cleanly converted to
Riparin 1.1b acid in a high isolated yield (71%) through the
NaNO2/MPAA/mercaptoethanol treatment. Under the
GSH/GSSG condition, Riparin 1.1b peptide acid was efficiently
folded into the desired Riparin 1.1b with an isolated yield of 65%.
Finally, we tested the utility of the new method for synthesis
of -conotoxin Vc1.1 containing two pairs of disulfide bonds.
This 16 amino acid peptide can alleviate neuropathinc pain in
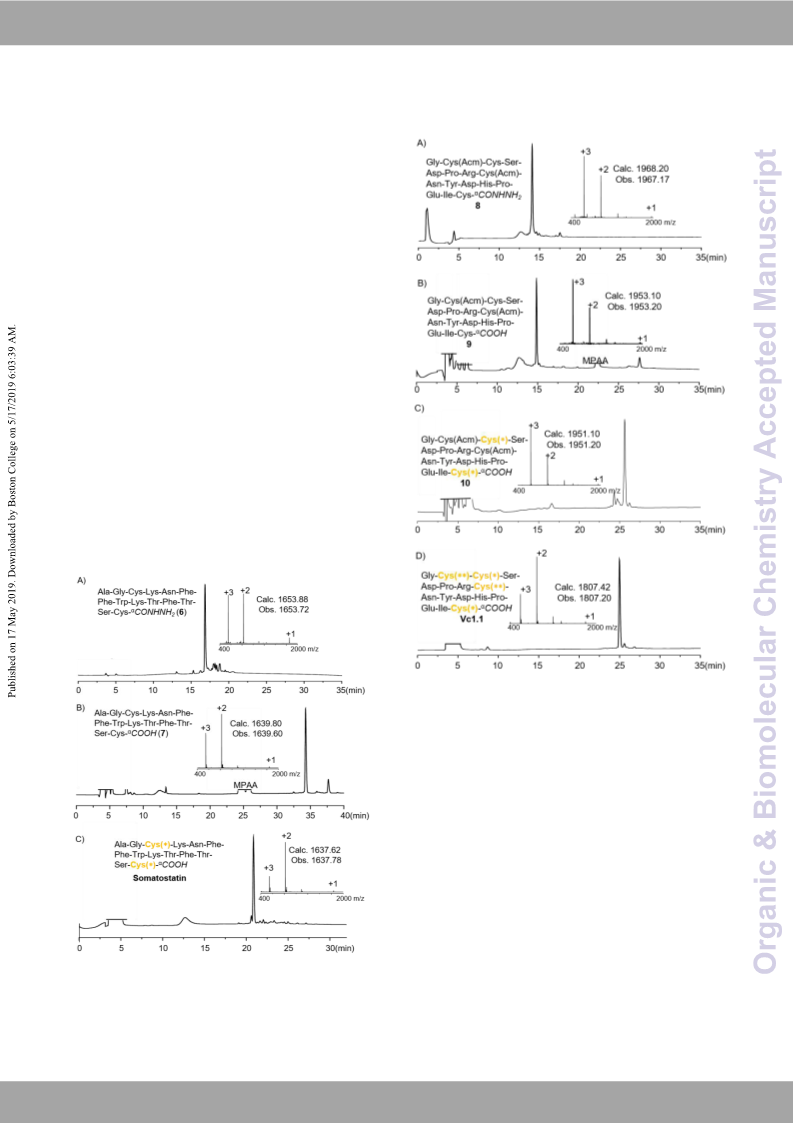
several rat models.14 The Vc1.1 hydrazide (8) was readily
prepared by Fmoc-based solid phase synthesis using hydrazine-
trityl(2-Cl) resin with an isolated yield of 68% (Fig. 5). After
sequential treatment with NaNO2, MPAA and mercaptoethanol,
this polypeptide hydrazide was almost quantitatively converted
to the unfolded Vc1.1 acid (9). Under the redox conditions of
GSH/GSSG, a disulfide bond was formed between Cys3 and
Cys16, affording 10 in a high analytical yield (82%). Upon
addition of I2, the side chain Acm-protecting group of Cys2 and
Cys8 was removed, followed by the formation of the second
disulfide bond (Fig. 5). The desired Vc 1.1 was isolated in 70%
yield.
To summarize, we have developed a new robust strategy for
the synthesis of C-terminal Cys-containing peptide acids. The
peptide hydrazide-based strategy has the following advantages:
1) C-terminal Cys hydrazide peptides can be readily prepared by
using low-cost hydrazine-trityl(2-Cl) resin; 2) Fmoc SPPS of C-
terminal Cys hydrazide peptides does not show any side
reactions of epimerization and -elimination at the C-terminal
Cys; 3) through the NaNO2/MPAA/mercaptoethanol treatment,
C-terminal hydrazide peptides can be cleanly converted to the
desired C-terminal Cys peptide acids. The utility of this method
has been demonstrated in the synthesis of C-AhPDF1.1b,
somatostatin, Riparian 1.1b, and -conotoxin Vc1.1 acid. Taken
together, the new method offers an efficient and practical
strategy for the preparation of C-terminal Cys peptide acids. Use
of the new method for the development of peptide
therapeutics and diagnositics is undergoing in our laboratory
and will be reported in due course.
4
5
6
7
8
F. Meindre, D. Lelièvre, K. Loth, O. Mith, V. Aucagne, P.
Berthomieu, L. Marquès, A. F. Delmas, F. Landon and F.
Paquet, Biochemistry 2014, 53, 7745-7754.
(a) J.-S. Zheng, S. Tang, Y.-K. Qi, Z.-P. Wang and L. Liu, Nat.
Protoc., 2013, 8, 2483-2495; (b) J.-S. Zheng, M. Yu, Y.-K. Qi, S.
Tang, F. Shen, Z.-P. Wang, L. Xiao, L. Zhang, C.-L. Tian and L.
Liu, J. Am. Chem. Soc., 2014, 136, 3695−3704; (c) Z. Wang, W.
Xu, L. Liu and T. F. Zhu, Nature Chem., 2016, 8, 698-704; (d) M.
Pan, S. Gao, Y. Zheng, X. Tan, H. Lan, X. Tan, D. Sun, L. Lu, T.
Wang, Q. Zheng, Y. Huang, J. Wang and L. Liu, J. Am. Chem.
Soc., 2016, 138, 7429-7435.
9
S. Tang, Y.-Y. Si, Z.-P. Wang, K.-R. Mei, X. Chen, J.-Y. Cheng, J.-
S. Zheng and L. Liu, Angew. Chem. Int. Ed., 2015, 54, 5713-
5717.
10 Z. P. Gates, J. R. Stephan, D. J. Lee and S. B. H. Kent, Chem.
Commun., 2013, 49, 786-788.
11 A. A. Vinogradov, M. D. Simon and B. L. Pentelute, Org. Lett.,
2016, 18, 1222-1225.
12 P. Brazeau, W. Vale, R. Burgus, N. Ling, M. Butcher, J. Rivier
and R. Guillemin, Science 1973, 179, 77–79.
13 V. M. Maselli, D. Bilusich, J. H. Bowie and M. J. Tyler, Rapid
Commun. Mass Spectrom., 2006, 20, 797-803.
14 (a) M. Vincler, S. Wittenauer, R. Parker, M. Ellison, B. M.
Olivera and J. M. McIntosh, Proc. Natl. Acad. Sci. USA 2006,
103, 17880-17884; (b) R. J. Clark, J. Jensen, S. T. Nevin, ; B. P.
Callaghan, D. J. Adams and D. J. Craik, Angew. Chem. Int. Ed.,
2010, 49, 6545-6548.
This work was supported by the National Key R&D Program
of China (2017YFA0505200), National Natural Science
Foundation of China (No. 21572214, 21807001, 91753205).
Conflicts of interest
There are no conflicts to declare.
Notes and references
1
(a) M. Góngora-Benítez, J. Tulla-Puche and F. Albericio, Chem.
Rev., 2014, 114, 901-926; (b) X. Xu and R. Lai, Chem. Rev., 2015,
4 | J. Name., 2012, 00, 1-3
This journal is © The Royal Society of Chemistry 20xx
Please do not adjust margins

 Zuo, Chao
Zuo, Chao
 Yan, Bing-Jia
Yan, Bing-Jia
 Zhu, Han-Ying
Zhu, Han-Ying
 Shi, Wei-Wei
Shi, Wei-Wei
 Xi, Tong-Kuai
Xi, Tong-Kuai
 Shi, Jing
Shi, Jing
 Fang, Ge-Min
Fang, Ge-Min