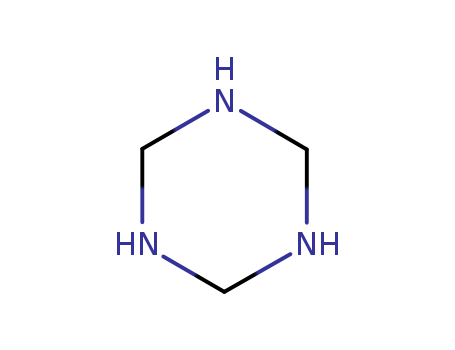
- Chemical Name:1,3,5-Triazinane
- CAS No.:110-90-7
- Molecular Formula:C3H9 N3
- Molecular Weight:87.1246
- Hs Code.:2933699090
- European Community (EC) Number:203-814-6
- UNII:M9XXT26VWH
- DSSTox Substance ID:DTXSID8059394
- Nikkaji Number:J193.585B
- Wikipedia:1,3,5-triazinane
- Wikidata:Q27117349
- Metabolomics Workbench ID:55751
- Mol file:110-90-7.mol
Synonyms:1,3,5-triazinane;cyclotrimethylenetriamine;110-90-7;Hexahydro-1,3,5-triazine;1,3,5-Triazine, hexahydro-;hexahydro-s-triazine;M9XXT26VWH;AI3-52753;EINECS 203-814-6;1,3,5-triazacyclohexane;trimethylenetriamine;UNII-M9XXT26VWH;S-TRIAZINE, HEXAHYDRO-;DTXSID8059394;CHEBI:38027;Q27117349