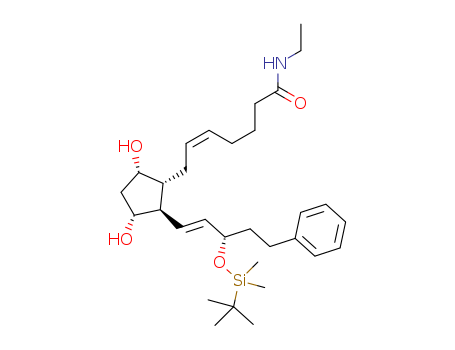
Technology Process of (3aR,4R,5R,6aS)-4-[(3R)-(tert-butyldimethylsilyl)oxy-5-phenylpentyl]perhydrocyclopenta[b]furan-2,5-diol
There total 10 articles about (3aR,4R,5R,6aS)-4-[(3R)-(tert-butyldimethylsilyl)oxy-5-phenylpentyl]perhydrocyclopenta[b]furan-2,5-diol which
guide to synthetic route it.
The literature collected by LookChem mainly comes from the sharing of users and the free literature resources found by Internet computing technology. We keep the original model of the professional version of literature to make it easier and faster for users to retrieve and use. At the same time, we analyze and calculate the most feasible synthesis route with the highest yield for your reference as below:
synthetic route:
- Guidance literature:
-
bimatoprost;
With
butylboronic acid;
In
dichloromethane;
for 0.666667h;
Reflux;
t-butyldimethylsiyl triflate;
With
2,6-dimethylpyridine;
In
dichloromethane;
at 20 ℃;
Cooling with ice;
- Guidance literature:
-
(5-(ethylamino)-5-oxopentyl)triphenylphosphonium bromide;
With
valerianate de potassium;
In
tetrahydrofuran;
at 0 ℃;
for 0.5h;
Inert atmosphere;
Schlenk technique;
(3aR,4R,5R,6aS)-4-[3S-(t-butyldimethylsiloxy)-5-phenyl-1E-pentenyl]-5-hydroxy-hexahydro-2H-cyclopenta[b]furan-2-ol;
In
tetrahydrofuran;
at 0 - 20 ℃;
for 4h;
Inert atmosphere;
Schlenk technique;
DOI:10.1021/ol503520f
- Guidance literature:
-
Multi-step reaction with 6 steps
1.1: sodium hydride / toluene; mineral oil / 20 h / Reflux
2.1: Schwartz's reagent / dichloromethane / 1 h / 20 °C / Inert atmosphere; Schlenk technique
2.2: 1 h / 0 - 20 °C / Inert atmosphere; Schlenk technique
3.1: tert.-butyl lithium / diethyl ether; pentane / 4 h / -78 - -40 °C / Inert atmosphere; Schlenk technique
3.2: 0.5 h / -30 °C / Inert atmosphere; Schlenk technique
4.1: ozone / methanol; dichloromethane / -78 °C
4.2: -78 - 20 °C
5.1: diisobutylaluminium hydride / dichloromethane / 2 h / -78 °C
6.1: valerianate de potassium / tetrahydrofuran / 0.5 h / 0 °C / Inert atmosphere; Schlenk technique
6.2: 4 h / 0 - 20 °C / Inert atmosphere; Schlenk technique
With
Schwartz's reagent; tert.-butyl lithium; valerianate de potassium; sodium hydride; diisobutylaluminium hydride; ozone;
In
tetrahydrofuran; methanol; diethyl ether; dichloromethane; toluene; mineral oil; pentane;
DOI:10.1021/ol503520f