
Chemistry - A European Journal p. 716 - 723 (2016)
Update date:2022-08-29
Topics:
 Chen, Xiaobei
Chen, Xiaobei
 Fan, Huaqiang
Fan, Huaqiang
 Zhang, Shilei
Zhang, Shilei
 Yu, Chenguang
Yu, Chenguang
 Wang, Wei
Wang, Wei
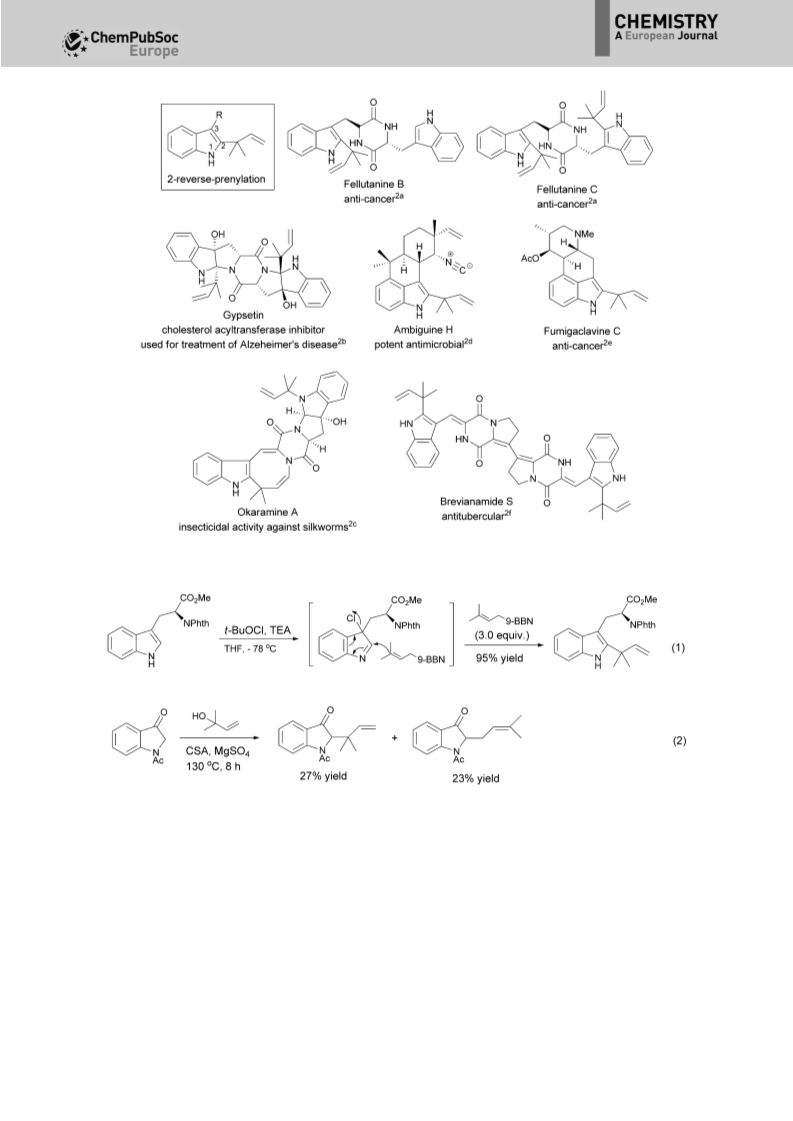
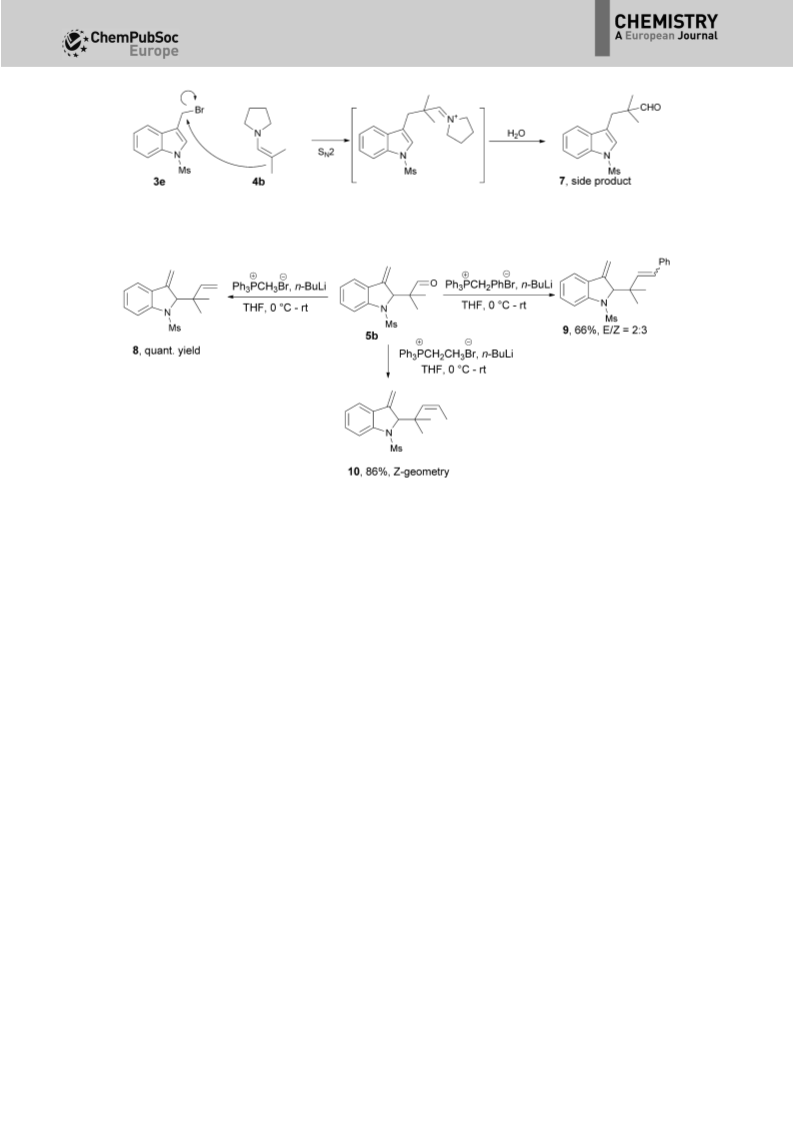
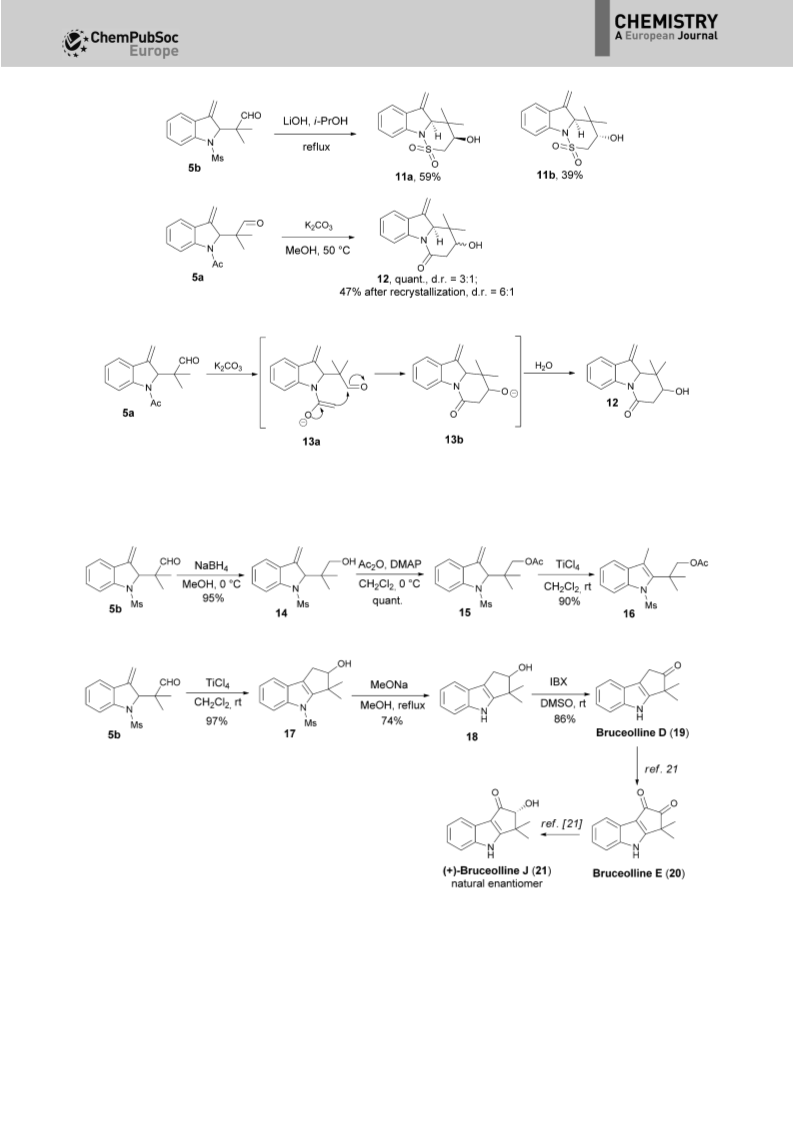
An unprecedented tandem N-alkylation-ionic aza-Cope (or Claisen) rearrangement-hydrolysis reaction of readily available indolyl bromides with enamines is described. Due to the complicated nature of the two processes, an operationally simple N-alkylation and subsequent microwave-irradiated ionic aza-Cope rearrangement-hydrolysis process has been uncovered. The tandem reaction serves as a powerful approach to the preparation of synthetically and biologically important, but challenging, 2-reverse quaternary-centered prenylated indoles with high efficiency. Notably, unusual nonaromatic 3-methylene-2,3-dihydro-1H-indole architectures, instead of aromatic indoles, are produced. Furthermore, the aza-Cope rearrangement reaction proceeds highly regioselectively to give the quaternary-centered reverse prenyl functionality, which often produces a mixture of two regioisomers by reported methods. The synthetic value of the resulting nonaromatic 3-methylene-2,3-dihydro-1Hindole architectures has been demonstrated as versatile building blocks in the efficient synthesis of structurally diverse 2-reverse prenylated indoles, such as indolines, indolefused sultams and lactams, and the natural product bruceolline D.
View More




Hubei Lansun Biochemical Pharmaceutical Co., Ltd
Contact:714-6395977
Address:No. 81 Pengcheng Avenue, economic and technological development zone, Huangshi City, Hubei Province,China
Chemtrade International ( China )
Contact:+86-532-86893005
Address:Rm 2-501, Huaxia Zonghe Building, No. 410 JInggangshan Road, Huangdao
Contact:+86-533-3112891
Address:zibo
Shanghai Forever Synthesis Co.,Ltd.
Contact:021-61124658
Address:Zhoukang Road,Pudong New District,Shanghai,China
Zhonghao (dalian) Research and Design Institute of Chemical Industry Co., Ltd
Contact:+86 411 84674606
Address:201, Huangpu Road , Shahekou District, Dalian ,116023-China
Doi:10.1246/cl.2005.474
(2005)Doi:10.1039/c0cc05018k
(2011)Doi:10.1016/S0040-4020(03)00342-9
(2003)Doi:10.1080/00397910008086916
(2000)Doi:10.3390/molecules26071925
(2021)Doi:10.1021/ol9908528
(1999)