
Organic Process Research and Development p. 301 - 305 (2020)
Update date:2022-08-22
Topics:
 Gao, Shenghua
Gao, Shenghua
 Gao, Xuezhi
Gao, Xuezhi
 Li, Houyong
Li, Houyong
 Yang, Anjiang
Yang, Anjiang
 Yang, Zhezhou
Yang, Zhezhou
 Zhang, Fuli
Zhang, Fuli
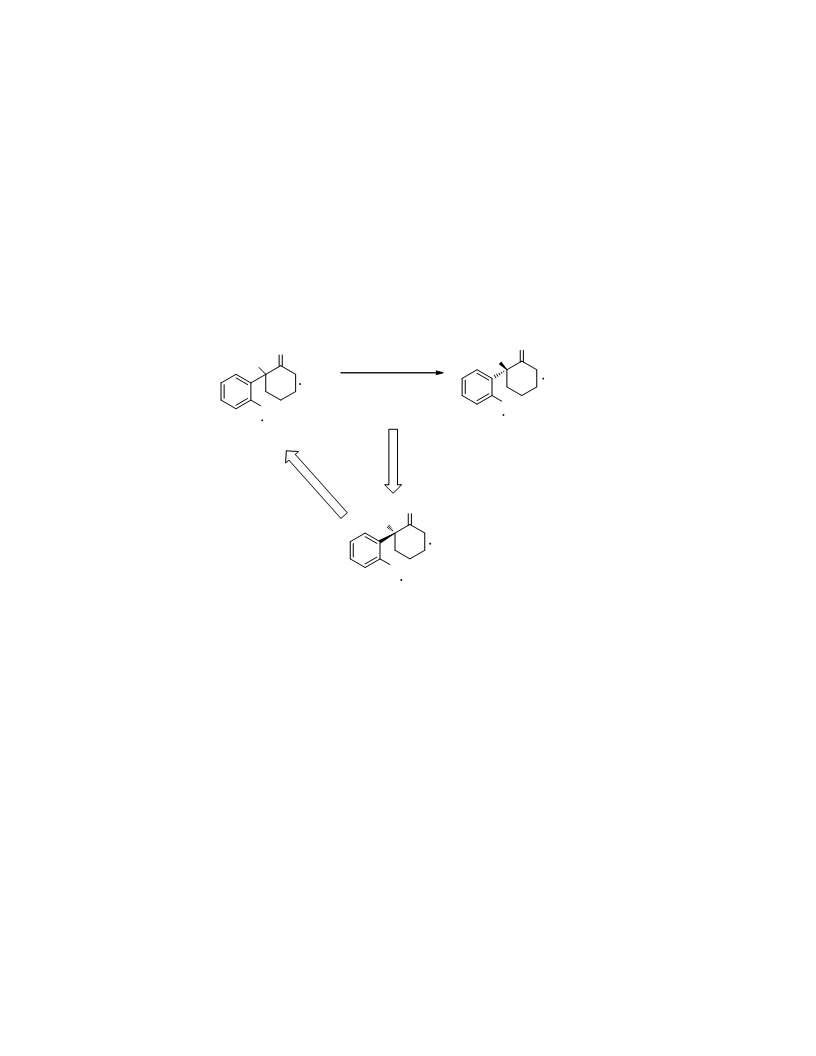
The first detailed description of the Lewis acid-catalyzed racemization of (R)-ketamine is reported. A process for racemization of the undesired (R)-ketamine enantiomer produced from the resolution for preparing the NMDA receptor antagonist (S)-ketamine was developed in quantitative yield with 99% chemical purity in the presence of a Lewis acid at 150 °C. Varying degrees of racemization were observed in the presence of various frequently used Lewis acids separately, and the catalytic efficiencies were arranged as follows: MgCl2 ≈ AlCl3 > FeCl3 > ZnCl2 > BF3 > CaCl2. The racemized ketamine was subsequently resolved using l-(+)-tartaric acid to obtain (S)-ketamine in 41% yield with 99.5% ee. Such a concise and cost-efficient approach for the racemization can be industrially useful to recycle the waste (R)-ketamine enantiomer into the resolution process to obtain (S)-ketamine.
View More










Doi:10.1021/ja01327a055
(1934)Doi:10.1055/s-2006-949606
(2006)Doi:10.1039/C29700000966
(1970)Doi:10.1016/j.mcat.2020.111009
(2020)Doi:10.1021/acs.jpca.6b01761
(2016)Doi:10.1039/c39940000549
(1994)